9908. Validamycins
Description and references
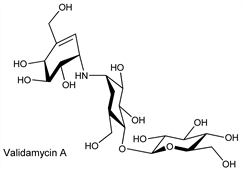
Antibiotic complex produced by Streptomyces hygroscopicus var limoneus. Consists of validamycins A (major component), B, C, D, E, and F. Isoln, characterization, and biological properties of validamycins A and B: Iwasa et al., J. Antibiot. 24, 107, 119 (1971). Isoln and characterization of validamycins C, D, E, F: Horii et al., ibid. 25, 48 (1972). Manuf: Horii et al., JP 72 39697 (1972 to Takeda), C.A. 78, 122657a (1973). Structure of validamycin A: Horii, Kameda, Chem. Commun. 1972, 747; revised structure: T. Suami et al., J. Antibiot. 33, 98 (1980). Bioassay methods: Iwasa et al., ibid. 24, 114 (1971). Total synthesis of (±)-validoxylamines A and B, constituents of validamycins: S. Ogawa et al., J. Org. Chem. 49, 2594 (1984). Total synthesis of validamycin B: S. Ogawa, Y. Miyamoto, Chem. Commun. 1987, 1843; of validamycin A: eidem, Chem. Lett. 1988, 889.