9962. Vernolepin
Nomenclature
CAS number: 18542-37-5
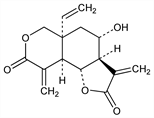
[3aR-(3aα,4α,5aα,9aα,9bβ)]-5a-Ethenyloctahydro-4-hydroxy-3,9-bis(methylene)-2H-furo[2,3-f][2]benzopyran-2,8(3H)-dione. C15H16O5; mol wt 276.28.
C 65.21%, H 5.84%, O 28.96%.
Description and references
The first recognized naturally occurring elemanolide dilactone; a sesquiterpene lactone with tumor inhibiting properties. Isoln from Vernonia hymenolepsis A. Rich, Compositae and structure: S. M. Kupchan et al., J. Am. Chem. Soc. 90, 3596 (1968); structural elucidation: eidem, J. Org. Chem. 34, 3903 (1969). Biological properties: S. M. Kupchan et al., J. Med. Chem. 14, 1147 (1971). Approach to stereosynthesis of (+)-form: H. Iio et al., Tetrahedron 35, 941 (1979); K. Kondo et al. Tetrahedron Lett. 34, 4219 (1993). Total synthesis of (±)-form: P. A. Grieco et al., J. Am. Chem. Soc. 98, 1612 (1976); F. Zutterman et al., Tetrahedron 35, 2389 (1979).
Properties
Crystals from chloroform-petr ether, mp 181-182°. [α]D28 +72° (c = 1.04 in acetone). uv max (methanol): 208 nm (ε 20300).Derivative
(±)-Form.Nomenclature
CAS number: 59598-29-7