9964. Verrucarins
Description and references
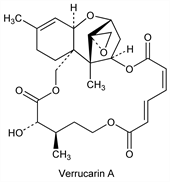
Macrocyclic tricothecane derivs which are secondary metabolites of the soil fungi Myrothecium verrucaria (Albertini et Schweinitz) Ditmar ex Fries; they are characterized by antibiotic, antifungal, and cytostatic activity. Verrucarins are triesters of the sesquiterpene alcohol verrucarol and closely related to the roridins, which are diesters of the same alcohol. Isoln of verrucarins A (major), B, C, D, E, F, G: E. Haerri et al., Helv. Chim. Acta 45, 840 (1962). Isoln of A: Symth, Kraskin, US 3087859 (1963 to Rohm & Haas); Vittimberga, J. Org. Chem. 28, 1786 (1963). Identity with muconomycin A: Vittimberga, Vittimberga, ibid. 30, 746 (1965). Structure of A: J. Gutzwiller, C. Tamm, Helv. Chim. Acta 48, 157 (1965). Stereochemistry: W. Zürcher et al., ibid. 840; A. T. McPhail, G. T. Sim, J. Chem. Soc. C 1966, 1394. Synthesis of A: W. C. Still, H. Ohmizu, J. Org. Chem. 46, 5242 (1981). Isoln of B: W. Loeffler et al., BE 627002 (1963 to Sandoz), C.A. 60, 10485b (1964). Structure of B: J. Gutzwiller, C. Tamm, Helv. Chim. Acta 48, 177 (1965). Toxicological studies of A and B: Guarino et al., Biotechnol. Bioeng. 10, 457 (1968); of A: Mortimer et al., Res. Vet. Sci. 12, 508 (1971). Biosynthetic studies of A and B: B. Müller, C. Tamm, Helv. Chim. Acta 58, 483 (1975); G. A. Cordell, Chem. Rev. 76, 425 (1976). Isoln of J (muconomycin B): Vittimberga, Vittimberga, loc. cit.; of H and J: B. Boehner et al., Helv. Chim. Acta 48, 1079 (1965). Structure of J: E. Fetz et al., ibid. 1669. Partial synthesis of tetrahydroverrucarin J and revised structure of J: W. Breitenstein, C. Tamm, ibid. 61, 1975 (1978). Isoln and structure of K: eidem, ibid. 60, 1522 (1977). Isoln of L: B. B. Jarvis et al., J. Antibiot. 34, 121 (1981). Total synthesis of verrucarol: R. H. Schlessinger, R. A. Nugent, J. Am. Chem. Soc. 104, 1116 (1982). Toxicity study: E. M. Rüsch, H. Sthlin, Arzneim.-Forsch. 15, 893 (1965). Comprehensive review of the verrucarins and roridins: C. Tamm, Fortschr. Chem. Org. Naturst. 31, 64-117 (1974). For a description of the stereochemistry of the roridins see B. R. Jarvis et al., J. Nat. Prod. 45, 440 (1982).