9976. Vidarabine
Nomenclature
Description and references
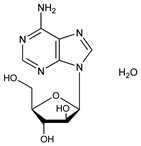
Purine nucleoside first synthesized as a potential anticancer agent: Lee et al., J. Am. Chem. Soc. 82, 2648 (1960); Reist et al., J. Org. Chem. 27, 3274 (1962); Glaudemans, Fletcher, ibid. 28, 3004 (1963); Reist et al., ibid. 29, 3725 (1964). Fermentation process using a strain of Streptomyces antibioticus: GB 1159290; J. D. Howells, A. Ryder, US 3616208 (1969, 1971 both to Parke, Davis). Crystal and molecular structure: Bunick, Voet, Acta Crystallogr. 30B, 1641 (1974). Series of articles on antiviral activity: Antimicrob. Agents Chemother. 1968, 136-179. Toxicity study: S. M. Kurtz et al., ibid. 180. HPLC determn in plasma and urine: W. P. McCann et al., ibid. 28, 265 (1985). Clinical trial in immunocompromised patients: R. J. Whitley et al., J. Infect. Dis. 165, 450 (1992). Book: Adenine Arabinoside: An Antiviral Agent, D. Paven-Langston et al., Eds. (Raven Press, New York, 1975) xviii + 425 pp. Review of pharmacology and clinical experience: R. A. Buchanan, F. Hess, Pharmacol. Ther. 8, 143-171 (1980). Comprehensive description: W. Hong et al., Anal. Profiles Drug Subs. 15, 647-672 (1986).
Properties
Crystals from water, mp 257.0-257.5° (0.4 H2O). [α]D27 5° (c = 0.25). uv max (pH 1): 257.5 nm (ε 12700); pH 7: 259 nm (ε 13400); pH 13: 259 nm (ε 14000). LD50 in mice (mg/kg): 4677 i.p.; >7950 orally (Kurtz).Therapeutic Category
Antiviral.
Keywords
Antiviral; Nucleosides/Nucleotides