9990. Vinorelbine
Nomenclature
Description and references
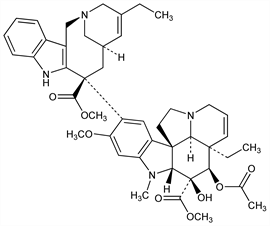
Semi-synthetic Vinca alkaloid; structurally related to vinblastine, q.v. Prepn: JP Kokai 80 31096; N. Langlois et al., US 4307100 (1980, 1981 both to Agence Nat. Valorisation Recherche); P. Mangeney et al., Tetrahedron 35, 2175 (1979). Pharmacology: G. Mathé, P. Reizenstein, Cancer Lett. 27, 285 (1985). HPLC determn in biological fluids: F. Jehl et al., J. Chromatogr. 525, 225 (1990). Veterinary trial in dogs with spontaneous neoplasia: V. J. Poirier et al., J. Vet. Intern. Med. 18, 536 (2004). Symposium: Semin. Oncol. 16, Suppl. 4, 1-45 (1989). Review of early clinical development: M. Marty et al., Nouv. Rev. Fr. Hematol. 31, 77-84 (1989); of clinical pharmacokinetics: D. Leveque, F. Jehl, Clin. Pharmacokinet. 31, 184-197 (1996); of use in advanced non-small cell lung cancer: M. P. Curran, G. L. Plosker, Drugs Aging 19, 695-721 (2002).
Properties
Residue. [α]D20 +52.4° (c = 0.3 in CHCl3). uv max (ethanol): 215, 268, 282, 293, 310 nm (ε 3700, 11000, 9500, 7600, 4400). Partition coefficient (octanol/buffer pH 7.2): 16. LD50 in mice (mg/m2): 72 i.v.; 78 orally (Mathé, Reizenstein).Derivative
Ditartrate.Nomenclature
Properties
Yellow-white amorphous powder. Sol in water and ethanol.Therapeutic Category
Antineoplastic.
Therapeutic Category (Veterinary)
Antineoplastic.Keywords
Antineoplastic; Alkaloids/Natural Products; Vinca Alkaloids