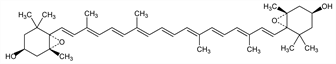
9998. Violaxanthin
Nomenclature
CAS number: 126-29-4
5,6:5′,6′-Diepoxy-5,5′,6,6′-tetrahydro-β,β-carotene-3,3′-diol; zeaxanthin diepoxide. C40H56O4; mol wt 600.87.
C 79.96%, H 9.39%, O 10.65%.
Description and references
Widely distributed carotenoid pigment. Formed in plants from zeaxanthin. Isoln from yellow pansies (Viola tricolor): Kuhn, Winterstein, Ber. 64, 326 (1931). Structure: Karrer et al., Helv. Chim. Acta 14, 1044 (1931); 16, 977 (1933); 19, 1024 (1936); 27, 1684 (1944). Partial synthesis: Karrer, Jucker, ibid. 28, 300 (1945). Abs config: Bartlett et al., J. Chem. Soc. C 1969, 2527. Isoln from Viola tricolor and configuration of the 15-cis-isomer: P. Molnar, J. Szabolcs, Phytochemistry 19, 623 (1980).