10003. Viquidil
Nomenclature
Description and references
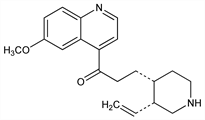
An isomer of quinine; occurs naturally as the d-form. Present in small quantities in cinchona barks; formed by heating quinine with glycerol at 180°: Howard, J. Chem. Soc. 24, 61 (1871); 25, 101 (1872); Miller, Rohde, Ber. 33, 3214 (1900); Howard, Chick, Pharm. J. 99, 143 (1917). Conversion to quinine: Rabe, Kindler, Ber. 51, 466 (1918). Partial synthesis: Prostenik, Prelog, Helv. Chim. Acta 26, 1965 (1943). Total synthesis: Woodward, Doering, J. Am. Chem. Soc. 66, 849 (1944); 67, 860 (1945); US 2500444 (1950 to Polaroid); Grethe et al., Helv. Chim. Acta 56, 1485 (1973). Review of synthesis, chemistry and pharmacology: Quevauviller et al., Ann. Pharm. Fr. 24, 39 (1966). Series of articles on pharmacology and metabolism: Arzneim.-Forsch. 22, 1334-1346 (1972).
Properties
Yellow viscous oil, [α]D +43°. Slightly sol in water; freely sol in alcohol, chloroform, ether.Derivative
Hydrochloride.Nomenclature
Properties
Yellow, odorless and bitter tasting powder, mp 184 ±4°. uv max (chloroform): 246, 355 nm. Sol in alc; sparingly sol in water. Practically insol in acetone.Therapeutic Category
Vasodilator (cerebral).
Keywords
Vasodilator (Cerebral)