10027. Voacamine
Nomenclature
Description and references
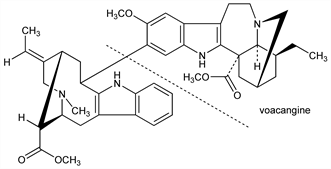
Bisindole alkaloid found in Voacanga africana Stapf., V. thouarsii R. & Sch., var obtusa (K. Sch.) Pichon and V. schweinfurthii Staph., Apocynaceae: Janot, Goutarel, Compt. Rend. 240, 1719 (1955); Rao, J. Org. Chem. 23, 1455 (1958); Janot, Goutarel, US 2823204 (1958 to Lab. Gobey). Identity with voacanginine: LeBarre, Gillo, Bull. Acad. R. Med. Belg. 20, 194 (1956). Cleaved by acid catalysis to voacangine: Winkler, Naturwissenschaften 48, 694 (1961). Structure: Goutarel et al., Compt. Rend. 243, 1670 (1956); Percheron, Ann. Chim. (Paris) 4, 303 (1959); Büchi et al., J. Am. Chem. Soc. 85, 1893 (1963). Review: Gorman et al. in The Alkaloids 1, J. E. Saxton, Ed. (The Chem. Soc., London, 1971) pp 242-249.
Properties
Prisms from acetone + methanol, dec 223°. [α]D20 52° (chloroform). uv max: 225, 295 nm (log ε 4.72, 4.28). Sol in chloroform, acetone; slightly sol in methanol, ethanol.Derivative
Voacangine.Nomenclature
Carbomethoxyibogaine; 12-methoxyibogamine-18-carboxylic acid methyl ester.Description and references
Structure: Bartlett et al., J. Am. Chem. Soc. 80, 126 (1958).