10031. Vomicine
Nomenclature
CAS number: 125-15-5
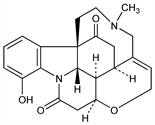
4-Hydroxy-19-methyl-16,19-secostrychnidine-10,16-dione; 12-hydroxy-N-methylpseudostrychnine. C22H24N2O4; mol wt 380.44.
C 69.46%, H 6.36%, N 7.36%, O 16.82%.
Description and references
From seed of Strychnos nux vomica L., Loganiaceae: Wieland, Oertel, Ann. 469, 193 (1929). Structure: Huisgen et al., ibid. 573, 121 (1951). Synthesis: Rosenmund, Angew. Chem. 75, 1127 (1963). Reviews: R. Robinson in Progress in Organic Chemistry vol. I (Butterworths, London, 1952) pp 2-21; J. B. Hendrickson in The Alkaloids vol. VI, R. H. F. Manske, H. L. Holmes, Eds. (Academic Press, New York, 1960) pp 195-204.
Properties
Hexagonal prisms from acetone, mp 284°. [α]D22 +80° (c = 0.5 in alc). Weak, mono-acidic base forming salts with an acid reaction. Freely sol in chloroform, sol in hot alcohol, acetone, slightly in ether, ethyl acetate.Derivative
Hydrochloride.C22H24N2O4.HCl; mol wt 416.90.
C 63.38%, H 6.04%, N 6.72%, O 15.35%, Cl 8.50%.
Properties
Prisms from water, dec 245°. uv max (ethanol): 222, 263, 297 nm (log ε 4.27, 3.77, 3.52). Sparingly sol in water.Derivative
Methyl ether.C23H26N2O4; mol wt 394.46.
C 70.03%, H 6.64%, N 7.10%, O 16.22%.