10109. Zalcitabine
Nomenclature
CAS number: 7481-89-2
2′,3′-Dideoxycytidine; dideoxycytidine; ddC; Hivid (Roche). C9H13N3O3; mol wt 211.22.
C 51.18%, H 6.20%, N 19.89%, O 22.72%.
Description and references
Pyrimidine nucleoside reverse transcriptase inhibitor. Prepn: J. P. Horwitz et al., J. Org. Chem. 32, 817 (1967); R. Marumoto, M. Honjo, Chem. Pharm. Bull. 22, 128 (1974). Prepn and structure-activity study: T.-S. Lin et al., J. Med. Chem. 30, 440 (1987). In vitro inhibition of HIV-1 virus replication: H. Mitsuya, S. Broder, Proc. Natl. Acad. Sci. USA 83, 1911 (1986). Review of pharmacology and clinical experience in HIV infection: G. Skowron, Adv. Exp. Med. Biol. 394, 257-269 (1996); J. C. Adkins et al., Drugs 53, 1054-1080 (1997).
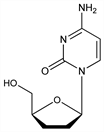
Properties
Crystals from ethanol + benzene, mp 215-217° (Horwitz); also reported as mp 209-210° (Lin). [α]D25 +81° (c = 0.635 in water). uv max in 0.1N HCl: 280 nm (ε 17720); in 0.1N NaOH: 270 nm (ε 8410). Soly in water (25°): 76.4 mg/ml.Therapeutic Category
Antiretroviral.
Keywords
Antiretroviral; Reverse Transcriptase Inhibitors; Nucleosides/Nucleotides; Reverse Transcriptase Inhibitor