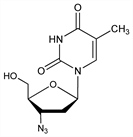
10123. Zidovudine
Nomenclature
Description and references
Pyrimidine nucleoside analog; reverse transcriptase inhibitor. Prepn: J. P. Horwitz et al., J. Org. Chem. 29, 2076 (1964); R. P. Glinski et al., ibid. 38, 4299 (1973). See also: J. L. Rideout et al., US 4724232 (1988 to Burroughs Wellcome). Total synthesis: C. K. Chu et al., Tetrahedron Lett. 29, 5349 (1988). HPLC determn and stability assay: A. Dunge et al., J. Pharm. Biomed. Anal. 37, 1109 (2005). In vitro antiviral and antitumor activity: E. De Clercq et al., Biochem. Pharmacol. 29, 1849 (1980); vs HIV-1 virus: H. Mitsuya et al., Proc. Natl. Acad. Sci. USA 82, 7096 (1985). Clinical pharmacokinetics: R. W. Klecker et al., Clin. Pharmacol. Ther. 41, 407 (1987). Toxicology studies: K. M. Ayers, Am. J. Med. 85, Suppl. 2A, 186 (1988). Comprehensive description: M. L. Sethi, Anal. Profiles Drug Subs. 20, 729-765 (1991). Review of clinical experience: G. X. McLeod, S. M. Hammer, Ann. Intern. Med. 117, 487-501 (1992); in prevention of perinatal HIV transmission: R. Sperling, Infect. Dis. Obstet. Gynecol. 6, 197-203 (1998).
Properties
Needles from petr ether, mp 106-112° (Horwitz). Also reported as crystals from water, mp 120-122° (Glinski). [α]D25 +99° (c = 0.5 in water). Soly in water (25°C): 25 mg/ml. uv max (water): 266.5 nm (ε 11650). LD50 in male, female mice, male, female rats (mg/kg): 3568, 3062, 3084, 3683 orally; >750 i.v. (all species) (Ayers).Therapeutic Category
Antiretroviral.
Therapeutic Category (Veterinary)
Antiviral in treatment of feline immunodeficiency virus and feline leukemia virus.Keywords
Reverse Transcriptase Inhibitor; Antiretroviral; Reverse Transcriptase Inhibitors; Nucleosides/Nucleotides