10165. Zineb
Nomenclature
Description and references
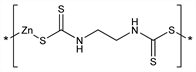
Polymeric salt of ethylenebisdithiocarbamic acid; related to maneb and mancozeb, q.q.v. Prepd from a zinc salt and nabam, q.v.: C. B. Luginbuhl, US 2690448 (1954 to du Pont). Fungicidal activity is due to degradation products, principally ethylenethiuram monosulfide: R. A. Ludwig et al., Can. J. Bot. 33, 42 (1955). Activity against potato blight: B. K. De, S. B. Chattopadhyay, Pesticides 18, 52 (1984). Decomposition of zineb to ethylenebisdicarbamate and ethylene thiourea, q.v., in tomatoes: B. D. Ripley, D. F. Cox, J. Agric. Food Chem. 26, 1137 (1978). HPLC determn: K. Gustafsson et al., ibid. 29, 729 (1981). Toxicity data: T. B. Gaines, Toxicol. Appl. Pharmacol. 14, 515 (1969). Aquatic toxicology: C. J. Van Leeuwen et al., Aquat. Toxicol. 7, 145 (1985).