10215. Prasugrel
Nomenclature
Description and references
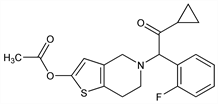
Thienopyridine antiplatelet prodrug. Deacetylated in vivo to an inactive, short-lived thiolactone metabolite; further metabolized by hepatic cytochrome P isoenzymes leading to the open ring, thiol-containing active metabolite, R-138727, which binds irreversibly to platelet P2Y12 adenosine diphosphate (ADP) receptors. Prepn: H. Koike et al., CA 2077695; eidem, US 5288726 (1993, 1994 to Sankyo). LC/MS/MS determn of metabolites in plasma: N. A. Farid et al., Rapid Commun. Mass Spectrom. 21, 169 (2007) DOI PubMed. Stereoselective antagonism in receptor-binding and platelet-aggregation assays: M. Hasegawa et al., Thromb. Haemostasis 94, 593 (2005) DOI. In vivo pharmacology: A. Sugidachi et al., Br. J. Pharmacol. 129, 1439 (2000) DOI PubMed. Clinical comparison with clopidogrel: M. Mariani et al., Expert Rev. Cardiovasc. Ther. 7, 17 (2009) DOI PubMed. Review of chemistry, metabolism, and clinical pharmacology: F. Asai et al., Annu. Rep. Sankyo Res. Lab. 51, 1-44 (1999); of clinical experience and therapeutic potential: U. S. Tantry et al., Expert Opin. Invest. Drugs 15, 1627-1633 (2006) DOI PubMed.
Properties
White to pale yellow crystalline powder containing masses. mp 121-122°. pKa (25°) 5.40±0.05. Log P (n-octanol/phosphate buffer): 3.7 (pH 7). Practically insol in water; becomes slightly sol with decreasing pH. Soly at 20° (mg/ml): benzene >100; chloroform >100; acetone >100; ethyl acetate 82.5; acetonitrile 67.0; methanol 9.01; ethanol 5.91; n-hexane 0.65. Soly at 20° (μg/ml): aq soln 59.8 (pH 3.57), 14.2 (pH 4.85), 4.01 (pH 6.00), 0.15 (pH 7.12).Derivative
Hydrochloride.Nomenclature
Derivative
R-138727.Nomenclature
Description and references
Active metabolite. Mixture of four stereoisomers; (4R,1S)-isomer identified as most active.
Therapeutic Category
Antithrombotic.
Keywords
Antithrombotic