10223. Romidepsin
Nomenclature
Description and references
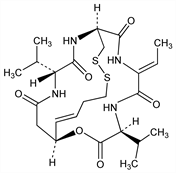
Bicyclic depsipeptide antibiotic produced by Chromobacterium violaceum WB968. Natural prodrug converted by glutathione-mediated reduction of the disulfide bond to the active histone deacetylase (HDAC) inhibitor. Isoln: M. Okuhara et al., EP 352646; eidem, US 4977138 (both 1990 to Fujisawa). Series of articles on isoln, structure, and activity: J. Antibiot. 47, 301-323 (1994) PubMed. Total synthesis: K. W. Li et al., J. Am. Chem. Soc. 118, 7237 (1996) DOI. HPLC determn in plasma: C. Chassaing et al., J. Chromatogr. B 719, 169 (1998) DOI PubMed. Mechanism of action studies: H. Nakajima et al., Exp. Cell Res. 241, 126 (1998) DOI PubMed; R. Furumai et al., Cancer Res. 62, 4916 (2002) PubMed. Clinical pharmacokinetics and toxicological evaluation: V. Sandor et al., Clin. Cancer Res. 8, 718 (2002) PubMed. Clinical evaluation in leukemias: J. C. Byrd et al., Blood 105, 959 (2005) DOI PubMed. Review of pharmacology and clinical experience: D. M. Vigushin, Curr. Opin. Investig. Drugs 3, 1396-1402 (2002) PubMed; P. A. Konstantinopoulos et al., Cancer Chemother. Pharmacol. 58, 711-715 (2006) DOI PubMed.
Properties
Colorless prisms, mp 235-245° (dec). [α]D23 +39° (c = 1.0 in CHCl3). Sol in chloroform, ethyl acetate; sparingly sol in methanol, ethanol. Insol in water, hexane.Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic; Histone Deacetylase Inhibitors