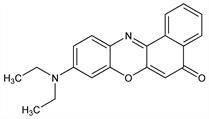
10229. Nile Red
Nomenclature
Description and references
Hydrophobic fluorescent and solvatochromic dye. Prepn: R. Mhlau, K. Uhlmann, Ann. 289, 90 (1896); J. F. Thorpe, J. Chem. Soc. 1907, 324. Solvent-dependent spectrophotometric properties: M. M. Davis, H. B. Hetzer, Anal. Chem. 38, 451 (1966) DOI. Spectrofluorometric studies of lipid interactions: P. Greenspan, S. D. Fowler, J. Lipid Res. 26, 781 (1985) PubMed. Solvatochromic and thermochromic properties: C. M. Golini et al., J. Fluoresc. 8, 395 (1998) DOI; N. Ghoneim, Spectrochim. Acta A 56, 1003 (2000) DOI PubMed. Use as a fluorescent probe of hydrophobic protein sites: D. L. Sackett, J. Wolff, Anal. Biochem. 167, 228 (1987) DOI PubMed; as a stain for low density lipoproteins: P. Greenspan, R. L. Gutman, Electrophoresis 14, 65 (1993) DOI PubMed; in detection of protein aggreagates: M. Sutter et al., J. Fluoresc. 17, 181 (2007) DOI PubMed; as an optochemical humidity sensor: I. Pellejero et al., Ind. Eng. Chem. Res. 46, 2335 (2007) DOI.