10232. Salinosporamides
Description and references
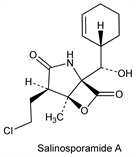
Family of cytotoxic proteasome inhibitors produced by the marine actinomycete, Salinispora tropica. Characterized by a fused γ-lactam-β-lactone ring structure; salinosporamide A is the most abundant. Isoln, structure, and bioactivity of A: R. H. Feling et al., Angew. Chem. Int. Ed. 42, 355 (2003) DOI PubMed; W. Fenical et al., US 7176232 (2007 to Regents of Univ. Calif.). Isoln of B and C: P. G. Williams et al., J. Org. Chem. 70, 6196 (2005) DOI PubMed; of D-J: K. A. Reed et al., J. Nat. Prod. 70, 269 (2007) DOI PubMed. Total syntheses of A: L. R. Reddy et al., J. Am. Chem. Soc. 126, 6230 (2004) DOI PubMed; A. Endo, S. J. Danishefsky, ibid. 127, 8298 (2005) DOI PubMed. Structure-activity study: V. R. Macherla et al., J. Med. Chem. 48, 3684 (2005) DOI PubMed. Pharmacology and antitumor activity of A: D. Chauhan et al., Cancer Cell 8, 407 (2005) DOI PubMed. Biosynthetic studies: G. Tsueng et al., Appl. Microbiol. Biotechnol. 75, 999 (2007) DOI PubMed; L. L. Beer, B. S. Moore, Org. Lett. 9, 845 (2007) DOI PubMed.
Derivative
Salinosporamide A.Nomenclature
Description and references
Soly and degradation study: N. Denora et al., J. Pharm. Sci. 96, 2037 (2007) DOI PubMed.