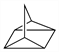
10239. Quadricyclane
Nomenclature
Description and references
Highly strained, thermally stable, saturated hydrocarbon. Prepn by photochemical isomerization of norbornadiene: W. G. Dauben, R. L. Cargill, Tetrahedron 15, 197 (1961) DOI; G. S. Hammond et al., J. Am. Chem. Soc. 83, 4674 (1961) DOI; C. D. Smith, Org. Synth. coll. vol. VI, 962 (1988). Cycloaddition reactions: M. E. Landis, J. C. Mitchell, J. Org. Chem. 44, 2288 (1979) DOI. Solar energy storage applications of photoisomerization reactions with norbornadiene: C. Philippopoulos et al., Ind. Eng. Chem. Prod. Res. Dev. 22, 627 (1983) DOI; K. Maruyama et al., J. Org. Chem. 50, 4742 (1985) DOI. Gas phase reactivity and thermochemistry: H. S. Lee et al., J. Am. Chem. Soc. 118, 5068 (1996) DOI. Vibrational spectroscopy: X. Zhou, R. Liu, Vib. Spectrosc. 12, 65 (1996) DOI. Review of isomerization reaction with norbornadiene and its energy storage applications: A. D. Dubonosov et al., Russ. Chem. Rev. 71, 917-927 (2002) DOI; of synthetic chemistry: V. A. Petrov, N. V. Vasil'ev, Curr. Org. Synth. 3, 215-259 (2006) DOI.