10252. Laquinimod
Nomenclature
Description and references
Immunomodulator. Prepn: A. Bjrk et al., WO 9955678; eidem, US 6077851 (1999, 2000 both to Active Biotech AB). Large scale synthesis: K. Jansson et al., J. Org. Chem. 71, 1658 (2006) DOI PubMed; J. Wennerberg et al., Org. Process Res. Dev. 11, 674 (2007) DOI. Structure activity studies: S. Jnsson et al., J. Med. Chem. 47, 2075 (2004) DOI PubMed. Determn in plasma by HPLC: K. Edman et al., J. Chromatogr. B 785, 311 (2003) DOI PubMed; by LC/MS/MS: C. J. Sennbro et al., Rapid Commun. Mass Spectrom. 20, 3313 (2006) DOI PubMed. Role of cytochrome P450 3A4 in metabolism: H. Tuvesson et al., Drug Metab. Dispos. 33, 866 (2005) DOI PubMed. Clinical evaluation in relapsing multiple sclerosis (MS): C. Polman et al., Neurology 64, 987 (2005) PubMed.
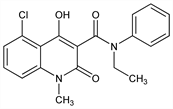
Properties
White to off-white crystals from n-heptane, mp 201° (dec). pKa 4.2.Derivative
Sodium salt.Nomenclature
Properties
White, non-hygroscopic crystals from ethanol.Therapeutic Category
Immunomodulator; in treatment of multiple sclerosis.
Keywords
Immunomodulator