10254. Plerixafor
Nomenclature
Description and references
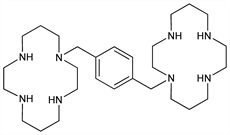
Symmetric bicyclam; specific CXCR4 chemokine receptor antagonist. Induces mobilization of CD34+ hematopoietic progenitor cells from bone marrow to peripheral blood. Prepn: M. Ciampolini et al., Inorg. Chem. 26, 3527 (1987) DOI; M. Achmatowicz, L. S. Hegedus, J. Org. Chem. 68, 6435 (2003) DOI PubMed; F. Boschetti et al., ibid. 70, 7042 (2005) DOI PubMed. Large scale synthesis: W. Yang et al., Tetrahedron Lett. 44, 2481 (2003) DOI. Receptor binding studies: M. M. Rosenkilde et al., J. Biol. Chem. 279, 3033 (2004) DOI PubMed; S. P. Fricker et al., Biochem. Pharmacol. 72, 588 (2006) DOI PubMed. Mechanism of action: H. E. Broxmeyer et al., J. Exp. Med. 201, 1307 (2005) DOI PubMed. Clinical pharmacokinetics and pharmacodynamics: N. A. Lack et al., Clin. Pharmacol. Ther. 77, 427 (2005) DOI PubMed. Clinical evaluation in mobilization of peripheral blood stem cells in multiple myeloma and non-Hodgkin's lymphoma: S. M. Devine et al., J. Clin. Oncol. 22, 1095 (2004) DOI PubMed. Review of discovery and clinical development: E. De Clercq, Nat. Rev. Drug Discovery 2, 581-587 (2003) DOI.
Properties
Colorless oil or foam.Derivative
Octahydrobromide.Nomenclature
Properties
Dihydrate as white solid, mp 239-241° (dec).Derivative
Octahydrochloride.Nomenclature
Properties
Trihydrate as fine colorless crystals.Therapeutic Category
Adjunct in bone marrow transplantation (stem cell mobilizer).