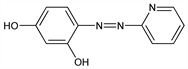
Nomenclature
CAS number: 1141-59-9;
113964-55-9
((E)-form)
4-[2-(2-Pyridinyl)diazenyl]-1,3-benzenediol; 4-(2-pyridinylazo)-1,3-benzenediol; 4-(2-pyridinylazo)resorcinol; 4-(2-pyridylazo)resorcinol.
C
11H
9N
3O
2; mol wt 215.21.
C 61.39%, H 4.22%, N 19.53%, O 14.87%.
Description and references
Terdentate ligand for cation chelation; reacts
with metal ions to form colored complexes. Prepn: A. E. Tschitschibabin, Chem.
Zentralbl. 1923, III, 1022. Improved synthesis: S. Wiejak et al., Pol. J. Chem. 58, 895 (1984). 1H NMR structural studies: K. Mochizuki et al., Bull. Chem.
Soc. Jpn. 52, 441 (1979) DOI. Acid-base properties and metal chelate
formation: T. Iwamoto, ibid. 34, 605 (1961) DOI. Absorption spectroscopy of PAR and its
metal chelates: W. J. Geary et al., Anal. Chim. Acta 26, 575 (1962) DOI. Stability constants of metal chelates:
eidem, ibid. 27, 71 (1962) DOI. Spectrophotometric determn of metal ions:
T. M. Florence, Y. Farrar, Anal. Chem. 35, 1613 (1963) DOI; E. Gómez et al., Fresenius J. Anal. Chem. 342, 318 (1992) DOI; M. N. Abbas et al., Anal. Chim. Acta 436, 223 (2001) DOI. Review of analytical uses: M. N. Desai, M. H. Gandhi, Rec. Chem. Progr. 30, 223-231 (1969).
Properties
Fine crystalline orange-red solid, mp 196-198° (Wiejak). Also reported
as amorphous brown solid from methanol, mp 182° (Geary). Irritant. pKa1 (25°) 5.83. pKa2 (25°) 12.5. Absorption max (free base,
pH 3.60): 383 nm (ε 15700);
(monoionic form, pH 5.98-12.50): 415 nm (ε 25900). Readily sol in aq alkaline
soln. Soly in water (10°): 5 mg/ml.Derivative
Sodium salt.
Nomenclature
CAS number: 13311-52-9;
16593-81-0
(monohydrate)
C
11H
8N
3NaO
2; mol wt 237.19.
C 55.70%, H 3.40%, N 17.72%, Na 9.69%, O 13.49%.
Properties
Sol in acids, alkalies, alcohols. Insol in ether.Use
Chromogenic indicator for complexometric, spectrophotometric,
and spot test detection and determn of metal ions. In ion chromatography
applications.
