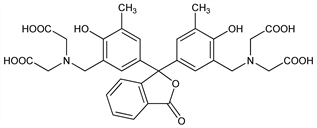
10274. o-Cresolphthalein Complexone
Nomenclature
Description and references
Cation chelating dye; forms 1:1 and 2:1 complexes with metal ions. Prepn from o-cresolphthalein, q.v.: G. Anderegg et al., Helv. Chim. Acta 37, 113 (1954) DOI; J. Krbl, R. Pribil, Chem. Ind. (London) 1957, 233. Spectrophotometric ionization studies: F. Gaizer et al., Talanta 28, 127 (1981) DOI; S. M. Abu-El-Wafa et al., Pak. J. Sci. Ind. Res. 28, 238 (1985). Polarography studies: X. Gao, M. Zhang, Anal. Chem. 56, 1912 (1984) DOI. Metal chelate studies: H. P. Srivastava, D. Tiwari, J. Chem. Eng. Data 41, 821 (1996) DOI; S. M. Abu-El-Wafa et al., J. Coord. Chem. 57, 813 (2004) DOI; T. M. Ismail et al., ibid. 1179 DOI; R. M. Issa et al., Synth. React. Inorg. Met.-Org. Chem. 34, 1087 (2005) DOI; K. Y. El-Baradie et al., Monatsh. Chem. 136, 1157 (2005) DOI. Determn of calcium in biological samples: G. Kessler, M. Wolfman, Clin. Chem. 10, 686 (1964) PubMed; B. C. Ray Sarkar, U. P. S. Chauhan, Anal. Biochem. 20, 155 (1967) DOI PubMed; K. Lorentz, Clin. Chim. Acta 126, 327 (1982) DOI PubMed; C. M. Corns, C. J. Ludman, Ann. Clin. Biochem. 24, 345 (1987) PubMed.