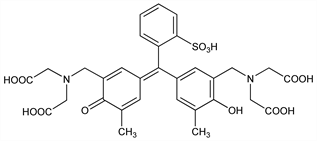
10279. Xylenol Orange
Nomenclature
Description and references
Cation chelant; sulfonephthalein titration indicator that forms soluble, colored complexes with metal ions. Exists in multiple sodium salt forms. Prepn from cresol red, q.v.: J. Krbl, R. Pribil, Chem. Ind. (London) 1957, 233; and chromatography: M. Murakami et al., Talanta 14, 1293 (1967) DOI; H. Nakayama et al., Anal. Sci. 5, 619 (1989) DOI. Polarography studies: M. Véber, L. J. Csányi, J. Electroanal. Chem. 101, 419 (1979) DOI. Ionization constant determn: M. B. Gholivand et al., Talanta 46, 875 (1998) DOI. Spectroscopy studies: S. Kiciak, H. Gontarz, ibid. 33, 341 (1986) DOI. Metal chelate studies: K. Ogura et al., J. Inorg. Nucl. Chem. 43, 1243 (1981) DOI. Hydroperoxide assay studies: C. Gay et al., Anal. Biochem. 273, 149 (1999) DOI PubMed; C. A. Gay, J. M. Gebicki, ibid. 315, 29 (2003) DOI PubMed; K. Arab, J.-P. Steghens, ibid. 325, 158 (2004) DOI PubMed. Use in fluorometric detection of calcified tissue: B. A. Rahn, S. M. Perren, Stain Technol. 46, 125 (1971) PubMed; Y.-H. Wang et al., Biotechnol. Prog. 22, 1697 (2006) PubMed; in fluorine determn: J. A. Ruzicka, et al., Talanta 13, 1341 (1966) DOI; in DNA determn: X. Chen et al., Spectrochim. Acta A 61, 2215 (2005) DOI PubMed. Reviews of analytical uses: B. Budesinsky in Chelates in Analytical Chemistry, H. A. Flaschka, A. J. Barnard, Jr., Eds. (Marcel Dekker, Inc., 1967) pp 15-47; S. Rani et al., Chem. Era 13, 372-388 (1978).