Nomenclature
CAS number: 3778-22-1
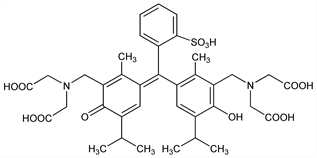
N,N′-[(1,1-Dioxido-3
H-2,1-benzoxathiol-3-ylidene)bis[[6-hydroxy-2-methyl-5-(1-methylethyl)-3,1-phenylene]methylene]]bis[
N-(carboxymethyl)glycine]; 3,3′-bis[
N,N-di(carboxymethyl)aminomethyl]thymolsulfonephthalein; thymolsulfonphthalein-3′,3″-bis(methyliminodiacetic acid).
C
37H
44N
2O
13S; mol wt 756.82.
C 58.72%, H 5.86%, N 3.70%, O 27.48%, S 4.24%.
Description and references
Multidentate cation chelant ligand; indicator
that forms soluble, colored complexes with metal ions. Prepn from thymol blue, q.v.: J. Krbl, R. Pribil, Chem.
Ind. (London) 1957, 233; J. Krbl, Collect. Czech. Chem. Commun. 22, 1789 (1957). Chromatographic purification: T.
Yoshino et al., Talanta 16, 151 (1969) DOI; S. Nakada et al., Bull. Chem. Soc. Jpn. 53, 3365 (1980) DOI. Spectroscopy studies: S. Kiciak, M. A. Mehdi, Talanta 39, 265 (1992) DOI; idem et
al., ibid. 42, 1245 (1995) DOI. Acid equilibrium and complex formation
constants: T. Yoshino et al., ibid. 21, 211 (1974) DOI. Metal chelate studies: B. Karadakov et al., ibid. 15, 525 (1968) DOI; C. Bremer, E.
Grell, Inorg. Chim. Acta 241, 13 (1996) DOI. Chelation ion chromatography applications:
B. Paull et al., Anal. Commun. 35, 17 (1998); eidem, Anal. Chim. Acta 375, 117 (1998) DOI. Reviews of analytical uses: B. Budesinsky
in Chelates in Analytical Chemistry, H. A. Flaschka, A. J. Barnard, Jr., Eds. (Marcel Dekker, Inc.,
1967) pp 15-47; S. Rani et al., Chem. Era 13, 372-388 (1978).
Properties
Solid, mp 252-256° (dec). Absorption max:
435 nm. pKa1 1.76;
pKa2 1.11; pKa3 0.78; pKa4 1.13; pKa5 2.60;
pKa6 3.24; pKa7 7.2; pKa8 11.1; pKa9 13.4. Color of aq soln transitions between yellow, blue, and violet;
color is dependent upon protonation state.Derivative
Tetrasodium salt.
Nomenclature
CAS number: 1945-77-3
C
37H
40N
2Na
4O
13S; mol wt 844.74.
C 52.61%, H 4.77%, N 3.32%, Na 10.89%, O 24.62%, S 3.80%.
Properties
Crystalline black powder of dull metallic luster.
Dec ≈300° with swelling. Sol in 2-3 parts cold water. Insol in
organic solvents. Absorption (0.5M sulfuric acid): 455
nm (ε 18900); 485 nm (ε 11800); 545 nm (ε 4450).Note
Not to be confused with methyl thymol
blue, a mixed acid-base indicator composed of methyl red and thymol
blue.Use
Chromogenic indicator for complexometric and spectrophotometric
determn of numerous metal ions, including iron, zinc, and magnesium.
Also to detect sulfate, fluoride, oxalate, and polyphosphate ions.
In chelation ion chromatography. As pH indicator.
