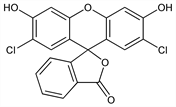
10288. 2′,7′-Dichlorofluorescein
Nomenclature
Description and references
Fluorescent dye. Prepn and use as argentometric titration indicator: I. M. Kolthoff et al., J. Am. Chem. Soc. 51, 3273 (1929) DOI. Dissociation and tautomerism: N. O. Mchedlov-Petrossyan et al., J. Phys. Org. Chem. 16, 380 (2003) DOI; and NMR spectroscopy: M. Afri et al., Chem. Phys. Lipids 131, 123 (2004) DOI PubMed. Acid-base equilibrium studies: H. Leonhardt et al., J. Phys. Chem. 75, 245 (1971) DOI. Chromatographic purification: W. A. Peeples II, J. R. Heitz, J. Liq. Chromatogr. 4, 51 (1981) DOI. Laser dye studies: R. W. Chambers, D. R. Kearns, Photochem. Photobiol. 10, 215 (1969) DOI PubMed; D. A. Jennings, A. J. Varga, J. Appl. Phys. 42, 5171 (1971) DOI; M. Yamashita et al., J. Chem. Phys. 66, 986 (1977) DOI. Prepn and applications of fluorescent derivatives: S. C. Burdette et al., J. Am. Chem. Soc. 123, 7831 (2001) DOI PubMed.