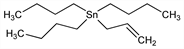
10295. Allyltributylstannane
Nomenclature
Description and references
Organotin reagent used to form carbon-carbon bonds. Prepn from allylmagnesium bromide and tri-n-butyltin chloride: W. J. Jones et al., J. Chem. Soc. 1947, 1446 DOI; from allylithium and tri-n-butyltin chloride: D. Seyferth, M. A. Weiner, J. Org. Chem. 26, 4797 (1961) DOI; from allylmagnesium bromide and bis(tributyltin) oxide: N. G. Halligan, L. C. Blaszczak, Org. Synth. coll. vol. VIII, 23 (1993). NMR studies: R. G. Jones et al., J. Organomet. Chem. 35, 291 (1972) DOI. Synthetic applications: M. Kosugi et al., Chem. Lett. 1977, 301; G. E. Keck, E. J. Enholm, J. Org. Chem. 50, 146 (1985) DOI; H. Nakamura et al., J. Am. Chem. Soc. 118, 6641 (1996) DOI; Y. Niwa, M. Shimizu, ibid. 125, 3720 (2003) DOI PubMed. Removal of organotin residues from reaction mixtures: C. J. Salomon et al., J. Org. Chem. 65, 9220 (2000) DOI PubMed. Review: A. J. Borah, Synlett 2008, 297-298 DOI; of allylstannanes: R. L. Marshall, Sci. Synth. 5, 573-605 (2003).