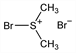
10307. Bromodimethylsulfonium Bromide
Nomenclature
Description and references
Brominating reagent in synthetic organic chemistry. Several forms of the dimethyl sulfide-bromine adduct exist; structure is dependent upon reaction conditions. Although the charge transfer-complex is more stable, the ionic species is often presumed when considering its synthetic applications. Prepn from dimethyl sulfide and bromine: A. Hantzsch, H. Hibbert, Ber. 40, 1508 (1907); P. Haas, Biochem. J. 29, 1297 (1935) PubMed; A. H. Fenselau, J. G. Moffatt, J. Am. Chem. Soc. 88, 1762 (1966) DOI; G. A. Olah et al., Synthesis 1979, 720 DOI; from DMSO and bromotrimethylsilane: G. Megyeri, T. Keve, Synth. Commun. 19, 3415 (1989) DOI; in situ from DMSO and aq hydrobromic acid: G. Majetich et al., J. Org. Chem. 62, 4321 (1997) DOI PubMed. Crystal and molecular structure studies: G. B. M. Vaughan et al., J. Chem. Soc. Dalton Trans. 1999, 79 DOI. Raman spectroscopy studies of structural forms: H. F. Askew et al., J. Raman Spectrosc. 22, 265 (1991) DOI. Synthetic applications in bromination reactions: G. Olah et al., Tetrahedron Lett. 20, 3653 (1979) DOI; Y. L. Chow, B. H. Bakker, Can. J. Chem. 60, 2268 (1982) DOI; in solvent free catalysis: A. T. Khan et al., J. Mol. Catal. A 239, 158 (2005) DOI; idem et al., Tetrahedron Lett. 48, 3805 (2007) DOI. Review: M. L. H. Choudhury, Synlett 2006, 1619-1620 DOI.