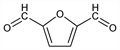
10310. 2,5-Furandicarboxaldehyde
Nomenclature
Description and references
Useful synthetic building block for the prepn of complex organic compds. Prepn from chloromethylfurfuraldehyde: W. F. Cooper, W. H. Nuttall, J. Chem. Soc. 101, 1074 (1912) DOI; from 2,5-bis(hydroxymethyl)furan: A. F. Oleinik, K. Y. Novitskii, J. Org. Chem. USSR (Engl. Transl.) 6, 2643 (1970); from (hydroxymethyl)furfural: E. L. Clennan, M. E. Mehrsheikh-Mohammadi, J. Am. Chem. Soc. 106, 7112 (1984) DOI; from fructose: G. A. Halliday et al., Org. Lett. 5, 2003 (2003) DOI PubMed. Synthetic applications: C. Domínguez et al., Synthesis 1989, 172; M. Badri et al., J. Am. Chem. Soc. 112, 5618 (1990) DOI; Z. Hui, A. Gandini, Eur. Polym. J. 28, 1461 (1992) DOI; A. Obrocka et al., Pol. J. Chem. 80, 1915 (2006). Review: J. Lewkowski, ARKIVOC 2001, 17-54.