10315. Belotecan
Nomenclature
Description and references
DNA topoisomerase I inhibitor; semi-synthetic analog of camptothecin, q.v. Prepd (not claimed): C. I. Hong et al., WO 9902530; eidem, US 6310207 (1999, 2001 both to Chong Kun Dang). Improved synthesis: S. K. Ahn et al., J. Heterocycl. Chem. 37, 1141 (2000). Characterization of physicochemical properties: J.-H. Kim et al., Int. J. Pharm. 239, 207 (2002) DOI PubMed. HPLC determn in plasma: J.-Y. Cho et al., J. Chromatogr. B 784, 25 (2003) DOI. Inhibition of topoisomerase I in mammalian tumor cells and human tumor xenografts: L.-H. Lee et al., Arch. Pharmacal Res. 21, 581 (1998). Clinical pharmacology: D. H. Lee et al., Clin. Cancer Res. 13, 6182 (2007) DOI PubMed. Clinical evaluation in small cell lung cancer: D. H. Lee et al., Ann. Oncol. 19, 123 (2008) DOI PubMed.
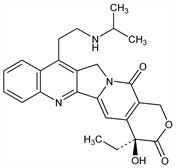
Derivative
Hydrochloride.Nomenclature
Properties
Slightly hygroscopic, pale yellow, crystalline powder, mp 267-268° (dec). [α]D +53.49° (c = 0.1 in water). pKa1 2.32±0.05, pKa2 9.15±0.02. Soly at 25° (mg/ml): acetic acid 10.70±0.08; methanol 4.13±0.32; ethanol 1.11±0.10; deionized water 8.22±0.18; acetonitrile 0.057±0.004.Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic; Alkaloids/Natural Products; Camptothecin Derivatives; Topoisomerase I Inhibitor