10321. Lasofoxifene
Nomenclature
Description and references
Selective estrogen receptor modulator (SERM). Partial estrogen agonist. Prepn: K. O. Cameron, P. A. Jardine, WO 9621656; K. O. Cameron et al., US 5552412 (1996, 1996 both to Pfizer). Structure-activity relationship: R. L. Rosati et al., J. Med. Chem. 41, 2928 (1998) DOI PubMed. Improved synthesis: X. Yang et al., Org. Lett. 2, 4025 (2000) DOI PubMed; Y. Sano et al., Chem. Lett. 36, 40 (2007) DOI. Crystal structure of complex with estrogen receptor α domain: F. F. Vajdos et al., Protein Sci. 16, 897 (2007) DOI PubMed. Clinical compaison with raloxifene, q.v., in postmenopausal osteoporosis: M. R. McClung et al., Menopause 13, 377 (2006) DOI PubMed. Review of preclinical studies: H. Z. Ke et al., J. Am. Aging Assoc. 25, 87-100 (2002) DOI; of pharmacology and clinical experience: L. Gennari et al., Expert Opin. Invest. Drugs 15, 1091-1103 (2006) DOI PubMed.
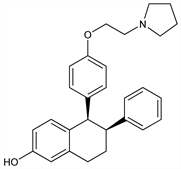
Derivative
Hydrochloride.Nomenclature
Properties
Crystals from acetonitrile/methylene chloride, mp 260-263°. [α]D 330.6° (c = 0.05 in CH2Cl2).Derivative
Tartrate.Nomenclature
Description and references
Prepn: C. K. Chiu, M. Meltz, US 5948809 (1999 to Pfizer). HPLC analysis of dissolution properties: J. S. Space et al., J. Pharm. Biomed. Anal. 44, 1064 (2007) DOI PubMed.
Properties
White solid from 95% ethanol.Therapeutic Category
Antiosteoporotic.
Keywords
Antiosteoporotic; Selective Estrogen Receptor Modulator (SERM)