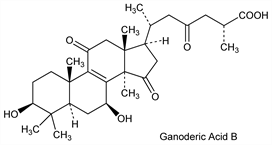
10350. Ganoderic Acids
Description and references
Family of bioactive, lanostane-type triterpenes isolated from the medicinal mushroom, Ganoderma lucidum (Fr.) Karst, Ganodermataceae. Numerous closely related structures have been identified that exhibit hepatoprotectant, antimicrobial, antihypertensive, antilipemic, or antitumor activities. Isoln and structures of ganoderic acids A and B: T. Kubota et al., Helv. Chim. Acta 65, 611 (1982) DOI; of ganoderic acids T through Z: J. O. Toth et al., Tetrahedron Lett. 24, 1081 (1983) DOI. Prodn by liquid fermentation: Q.-H. Fang, J.-J. Zhong, Biotechnol. Prog. 18, 51 (2002) DOI PubMed. Structural analysis and bioactivities: M.-S. Shiao et al., ACS Symp. Ser. 547, 342 (1994). Quantitative determn of major components by HPLC: X.-M. Wang et al., J. Pharm. Biomed. Anal. 41, 838 (2006) DOI PubMed. Analysis in chloroform extracts of G. lucidum by HPLC/ESI-MS: M. Yang et al., J. Am. Soc. Mass Spectrom. 18, 927 (2007) DOI PubMed. HPLC determn in plasma: X. Wang et al., Biomed. Chromatogr. 21, 389 (2007) DOI PubMed. Review of structures and properties: R. J. Cole, M. A. Schweikert, Handbook of Secondary Fungal Metabolites Vol. 2 (Academic Press, San Diego, 2003) pp 271-350. Review: Y. Zhou et al., Food Rev. Int. 22, 259-273 (2006) DOI.