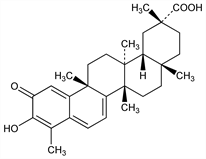
10370. Celastrol
Nomenclature
Description and references
Anti-inflammatory triterpenoid isolated from the Chinese medicinal herb, thunder god vine, Tripterygium wilfordii Hook. f., and other plants of the family, Celastraceae. Identification as red pigment in T. wilfordii: T. Q. Chou, P. F. Mei, Chin. J. Physiol. 10, 529 (1936); eidem, Chin. Med. J. 54, 37 (1938). Isoln from Celastrus scandens: O. Gisvold, J. Am. Pharm. Assoc. 28, 440 (1939). Identity of tripterine and celastrol: M. S. Schechter, H. L. Haller, J. Am. Chem. Soc. 64, 182 (1942) DOI. Structure: R. Harada et al., Tetrahedron Lett. 3, 603 (1962) DOI. Isoln from Reissantia buchananii: F.-R. Chang et al., J. Nat. Prod. 66, 1416 (2003) DOI PubMed. HPLC/MS/MS determn in human blood: Q. Xu et al., Chromatographia 66, 735 (2007) DOI. Antiangiogenic and antitumor activity: Y. Huang et al., Cancer Lett. 264, 101 (2008) DOI PubMed. Efficacy in rat models of arthritis: H. Li et al., J. Ethnopharmacol. 118, 479 (2008) DOI PubMed.