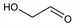10382. Glycolaldehyde
Nomenclature
Description and references
Simplest possible monosaccharide; isomer of methyl formate and acetic acid, q.q.v. Detected in interstellar space and thought to be key to the prebiotic formation of sugars and amino acids. Exists as a dimer in solid form, dissociates into the monomer in solution. Prepn from dihydroxymaleic acid: H. J. H. Fenton, H. Jackson, J. Chem. Soc. 75, 575 (1899) DOI; from divinyl ether: N. A. Milas et al., J. Am. Chem. Soc. 61, 1844 (1939) DOI; from ethylene glycol: H. Ukeda et al., Biosci. Biotechnol. Biochem. 62, 1589 (1998) DOI. Dissociation of dimer in soln: N. P. McCleland, J. Chem. Soc. 99, 1827 (1911) DOI; R. P. Bell, J. P. H. Hirst, ibid. 1939, 1777 DOI; C. I. Stassinopoulou, C. Zioudrou, Tetrahedron 28, 1257 (1972) DOI. Autocatalytic role in the production of sugars from formaldehyde (formose reaction): R. Breslow, Tetrahedron Lett. 1, No. 21, 22 (1959) DOI. Use in organic synthesis: P. Dinprasert et al., Tetrahedron Lett. 30, 1149 (1989) DOI; in polyol synthesis of silver nanostructures: S. E. Skrabalak et al., Nano Lett. 8, 2077 (2008) DOI PubMed. Role in prebiotic synthesis of amino acids: G. Zubay, Chemtracts Biochem. Mol. Biol. 10, 407 (1997); of carbohydrates: O. P. Pestunova et al. in Biosphere Origin and Evolution (Springer-Verlag, Heidelberg, 2008) pp 103-117. Detection in interstellar clouds: J. M. Hollis et al., Astrophys. J. 540, L107 (2000) DOI; outside the galactic center: M. T. Beltrán et al., ibid. 690, L93 (2009) DOI.