10395. NB-Enantride
Nomenclature
CAS number: 81572-37-4
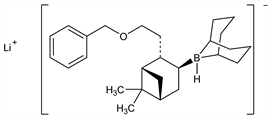
(T-4)-1,5-Cyclooctanediyl[(1S,2S,3S,5R)-6,6-dimethyl-2-[2-(phenylmethoxy)ethyl]bicyclo[3.1.1]hept-3-yl]hydroborate(1)
lithium; lithium hydrido(9-BBN-nopol benzyl ether adduct). C26H40BLiO; mol wt 386.35.
C 80.83%, H 10.44%, B 2.80%, Li 1.80%, O 4.14%.
Description and references
Lithium trialkylborohydride reagent for the asymmetric reduction of ketones. Prepn and synthetic applications: M. M. Midland, A. Kazubski, J. Org. Chem. 47, 2495 (1982) DOI. Reactivity comparison with other asymmetric reducing agents: H. C. Brown et al., ibid. 52, 5406 (1987) DOI; M. M. Midland et al., ibid. 56, 1068 (1991) DOI; S. A. Weissman, P. V. Ramachandran, Tetrahedron Lett. 37, 3791 (1996) DOI.
Properties
Sol in tetrahydrofuran. Flammable. Irritant. Reacts violently with water.Derivative
NB-Enantrane.Nomenclature
CAS number: 81971-15-5
9-[(1S,2R,3S,5S)-6,6-Dimethyl-2-[2-(phenylmethoxy)ethyl]bicyclo[3.1.1]hept-3-yl]-9-borabicyclo[3.3.1]nonane; 9-BBN-nopol benzyl ether adduct. C26H39BO; mol wt 378.40.
C 82.53%, H 10.39%, B 2.86%, O 4.23%.
Description and references
Precursor to NB-enantride; also utilized for asymmetric ketone reductions. Prepn from nopol benzyl ether and synthetic applications: M. M. Midland, A. Kazubski, J. Org. Chem. 47, 2814 (1982) DOI. Synthetic limitations: H. C. Brown et al., ibid. 55, 6328 (1990) DOI.