12-01234. Betonicine [Archived]
Nomenclature
CAS number: 515-25-3
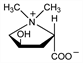
(2S-trans)-2-Carboxy-4-hydroxy-1,1-dimethylpyrrolidinium
inner salt; l-N,N-dimethyl-4-hydroxypyrrolidine-2-carboxylic
acid betaine; l-1-methyl-4-hydroxypyrrolidine-2-carboxylic acid
methylbetaine; l-4-hydroxystachydrine; l-4-hydroxyproline betaine. C7H13NO3; mol wt 159.18.
C 52.82%, H 8.23%, N 8.80%, O 30.15%.
Description and references
Occurs in Croton gubouga S. Moore; in Stachys officinalis (L.) Trev. (Betonica officinalis L.), Labiatae; in Achillea moschata Jacq. and A. millefolium L., Compositae: Goodson, Clewer, J. Chem. Soc. 115, 923 (1919); Guggenheim, Die biogenen Amine (S. Karger, New York, 1951) p 246; Miller, Chow, J. Am. Chem. Soc. 76, 1353 (1954); Pailer, Kump, Monatsh. Chem. 90, 396 (1959); Arch. Pharm. 293, 646 (1960). Stereoisomeric with turicine (cis-form). Synthesis: Patchett, Witkop, J. Am. Chem. Soc. 79, 185 (1957).
Properties
Blunt prisms from ethanol, dec 254-256°. Sweet taste. [α]D20 34.2° (c = 1.0). Readily sol in water or hot alcohol, slightly in cold alcohol. Practically insol in benzene, ether, chloroform, carbon tetrachloride.Derivative
Hydrochloride.C7H13NO3.HCl; mol wt 195.64.
C 42.97%, H 7.21%, N 7.16%, O 24.53%, Cl 18.12%.
Properties
Crystals from ethanol, dec 216-217° (after drying). [α]D20 24.2° (c = 0.0892 in water).Derivative
Aurichloride.C7H13NO3.HAuCl4; mol wt 498.97.
C 16.85%, H 2.83%, N 2.81%, O 9.62%, Au 39.47%, Cl 28.42%.
Properties
Scaly clusters from water, mp 242°.Status
This monograph has been retired and is no longer subject to revision or update.