12-03634. Enteromycin [Archived]
Nomenclature
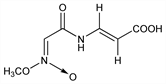
CAS number: 3552-16-7
3-[[(Methyl-aci-nitro)acetyl]amino]-2-propenoic
acid; 3-[2-(methyl-aci-nitro)acetamido]acrylic acid; N-(O-methyl-aci-nitroacetyl)-3-aminoacrylic
acid; seligocidin. C6H8N2O5; mol wt 188.14.
C 38.30%, H 4.29%, N 14.89%, O 42.52%.
Description and references
Antibiotic substance isolated from Streptomyces albireticuli: Nakazawa, C.A. 51, 10647i (1957); from S. achromogenes: Herr et al., Antimicrob. Agents Chemother. 1964, 100. Structure: Mizuno, Bull. Chem. Soc. Jpn. 34, 1419, 1425, 1631, 1633 (1961). Process for production: Shibata et al., JP 58 5747 (1958 to Takeda), C.A. 53, 16481f (1959). Identification of carboxamide: S. E. DeVoe et al., Antimicrob. Agents Chemother. 1964, 105; L. A. Mitscher et al., Tetrahedron 21, 267 (1965).
Properties
Two interchangeable crystal forms from methanol: needles or fine prisms. Indefinite mp. pKa 4.3. Unstable to heat, acid and alkali. Sparingly sol in water and other solvents. uv max (methanol): 230, 275, 298 nm (ε 9000, 13000, 16000).Derivative
Carboxamide.C6H9N3O4; mol wt 187.15.
C 38.51%, H 4.85%, N 22.45%, O 34.20%.
Properties
Rosettes from glacial acetic acid, dec 155°. uv max (methanol): 230, 300 nm (E1%1cm 650, 820). Sol in glacial acetic acid, dimethyl sulfoxide, dimethylformamide; slightly sol in methanol. Practically insol in the usual organic solvents.Derivative
Methyl ester.C7H10N2O5; mol wt 202.16.
C 41.59%, H 4.99%, N 13.86%, O 39.57%.
Properties
Crystals from ethanol, mp 141°.Status
This monograph has been retired and is no longer subject to revision or update.