12-06616. Nidulin [Archived]
Nomenclature
CAS number: 10089-10-8
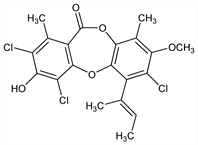
(E)-2,4,7-Trichloro-3-hydroxy-8-methoxy-1,9-dimethyl-6-(1-methyl-1-propenyl)-11H-dibenzo[b,e][1,4]dioxepin-11-one; methylustin; ustin methyl ether. C20H17Cl3O5; mol wt 443.70.
C 54.14%, H 3.86%, Cl 23.97%, O 18.03%.
Description and references
Antibiotic substance produced by a strain of Aspergillus nidulans (Eidam) Wint.: Dean et al., Nature 172, 344 (1953); Dean et al., J. Chem. Soc. 1954, 1432. Structure: Dean et al., ibid. 1960, 4829; Bycroft et al., ibid. 1963, 5148.
Properties
Slender, shiny rods from petr ether, mp 180°. Practically insol in water. Sol in aq solns of sodium hydroxide and sodium bicarbonate. Freely sol in chloroform. Sparingly sol in 95% ethanol and in benzene. Very sparingly sol in hot petr ether. A 4% soln in chloroform shows no optical activity. Completely inhibits in vitro the growth of Mycobacterium tuberculosis for four weeks at a diln of between 1 in 5000 and 1 in 10,000 when tested in the presence of serum by modified Long's medium and the floating-pellicle method. A 0.1% soln also inhibits the growth of the human parasitic fungi Trichophyton tonsurans and Microsporum audouini, but shows little or no activity towards a wide range of other microorganisms. Inactive against bacteriophage.Status
This monograph has been retired and is no longer subject to revision or update.