12-08522. Sarmentogenin [Archived]
Nomenclature
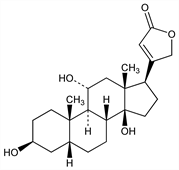
CAS number: 76-28-8
(3β,5β,11α)-3,11,14-Trihydroxy-card-20(22)-enolide. C23H34O5; mol wt 390.51.
C 70.74%, H 8.78%, O 20.49%.
Description and references
An aglucon from sarmentocymarin isolated from the seeds of Strophanthus sarmentosus DC. var. senegambiae (A. DC.) Monachino, Apocynaceae: Jacobs, Heidelberger, J. Biol. Chem. 81, 765 (1929); Katz, Helv. Chim. Acta 31, 993 (1948). Structure: Callow, Taylor, J. Chem. Soc. 1952, 2299; Repke, Klesczewski, Arch. Exp. Pathol. Pharmakol. 239, 131 (1960).
Properties
Prisms from methanol or methanol + acetone, mp 278-282°. [α]D19 +21.1 ± 4° (c = 0.521 in methanol). Absorption max (98% H2SO4): 230, 415 nm (E1%1cm 460, 430). Sol in alcohol, methanol, pyridine; sparingly sol in acetone, chloroform; practically insol in benzene, ether. Dissolves in concd H2SO4 with a bright golden color which slowly turns green and then indigo. In dil pyridine soln gives a deep red color with alkaline nitroprusside soln. When recrystallized from pyridine, it forms long microplatelets contg ≈1 mol of the solvent and melting at 258° with effervescence. Is not precipitated with digitonin. Yields sarmentogenone on oxidation.Derivative
Diacetate.C27H38O7; mol wt 474.59.
C 68.33%, H 8.07%, O 23.60%.
Properties
Needles from abs ether, mp 135-155°. Not affected by CrO3 in acetic acid.Derivative
Dibenzoate.C37H42O7; mol wt 598.73.
C 74.22%, H 7.07%, O 18.71%.
Properties
Flat hexagonal prisms from acetone, mp 281°. [α]D20 +14° (acetone). Sparingly sol in alc; practically insol in ether. Not affected by CrO3 in acetic acid.Status
This monograph has been retired and is no longer subject to revision or update.