12-09425. Thevetose [Archived]
Nomenclature
CAS number: 5675-98-9
6-Deoxy-3-O-methylglucose; 3-O-methylglucomethylose; quinovose 3-methyl ether. C7H14O5; mol wt 178.18.
C 47.19%, H 7.92%, O 44.90%.
Description and references
Isoln of l-thevetose from Thevetia nereifolia Juss. and Tanghinia venenifera Poir., Apocynaceae: Frèrejacque, Compt. Rend. 221, 645 (1945); Frèrejacque, Hasenfratz, ibid. 222, 815 (1946); Sigg et al., Helv. Chim. Acta 38, 166 (1955); from thevetin: Helfenberger, Reichstein, ibid. 31, 1470 (1948); from Bowiea volubilis Harv., Liliaceae: Tschesche, Dlberg, Ber. 90, 2378 (1957). Synthesis: Blindenbacher, Reichstein, Helv. Chim. Acta 31, 1669 (1948). Isoln of d-thevetose from Adenium honghel D.C., Apocynaceae: Frèrejacque, Compt. Rend. 230, 127 (1950). Rare instance of two optical isomers of the same sugar being present in the vegetable kingdom.
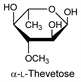
Derivative
l-Form.Properties
Needles from acetone + ether, mp 128-130°. [α]D19 66 to 36.9° (c = 1.46 in water).Derivative
β-l-Triacetate.C13H20O8; mol wt 304.29.
C 51.31%, H 6.62%, O 42.06%.
Properties
Crystals, mp 118-119°. [α]D18 7.5° (c = 1.12 in acetone).Derivative
l-Thevetosazone.Properties
Crystals, mp 136-139°. [α]D24 +110.8 to +40.3°.Derivative
d-Form.Properties
Crystals, mp 116°. [α]D20 +84 to +33°.Derivative
β-d-Triacetate.C13H20O8; mol wt 304.29.
C 51.31%, H 6.62%, O 42.06%.
Properties
Crystals, mp 121°. [α]D20 +6° (acetone).Derivative
α-d-Triacetate.Properties
Crystals, mp 105°. [α]D20 +122° (acetone).Derivative
d-Thevetosazone.Properties
Crystals, mp 123-128°. [α]D16 117.8 to 26.4°.Status
This monograph has been retired and is no longer subject to revision or update.