13-04337. Fuscin [Archived]
Nomenclature
CAS number: 83-85-2
9,10-Dihydro-5-hydroxy-4,8,8-trimethyl-2H,4H-benzo[1,2-b:4,3-c′]dipyran-2,6(8H)-dione. C15H16O5; mol wt 276.28.
C 65.21%, H 5.84%, O 28.96%.
Description and references
Antibacterial pigment produced by the fungus Oidiodendron fuscom Robak: Michael, Biochem. J. 42, XL; 43, 528 (1948). Derivatives and degradation products: Birkinshaw et al., ibid. 48, 66 (1951). Structure and synthesis: Birch, Chem. Ind. (London) 1955, 682; Barton, Hendrickson, J. Chem. Soc. 1956, 1028. Alternate synthesis: Pyuskyulev et al., Tetrahedron 29, 2849 (1973). Biosynthesis: Birch, Ciba Found. Symp. Quinones Electron Transp. 1960, 233; Birch et al., J. Chem. Soc. 1965, 1231.
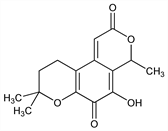
Properties
Orange-colored, diamond-shaped plates from alc, mp 230°. uv max: 355, 283 nm (ε 27,600; 1000). Practically insol in water, petr ether. Sol in chloroform, acetone, ethyl acetate, acetic acid, ether, benzene. Slightly sol in cold ethanol, more sol in hot ethanol. Dissolves in aq solns of alkali hydroxides and pyridine with an intense purple color. Easily reduced by hydrosulfite or other reducing agents to the colorless dihydrofuscin, C15H18O5, mp 206°, which is also found in the metabolic soln from the fungus grown on Czapek-Dox medium.Status
This monograph has been retired and is no longer subject to revision or update.