13-06304. Morphothebaine [Archived]
Nomenclature
CAS number: 478-53-5
5,6,6a,7-Tetrahydro-10-methoxy-6-methyl-4H-dibenzo[de,g]quinoline-2,11-diol; 10-methoxyaporphine-2,11-diol; 2,11-dihydroxy-10-methoxyaporphine. C18H19NO3; mol wt 297.35.
C 72.71%, H 6.44%, N 4.71%, O 16.14%.
Description and references
Prepd by heating thebaine with HCl. Formation from bulbocapnine: Ayer, Taylor, J. Chem. Soc. 1956, 472. Review: Small, Lutz, “Chemistry of the Opium Alkaloids” in U.S. Public Health Reports Suppl. No. 103 (Washington, 1932); R. H. F. Manske, H. L. Holmes, The Alkaloids vol. II (Academic Press, New York, 1952) pp 112-116.
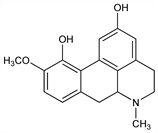
Properties
Rhombic crystals, dec 197°. [α]D15 130° (c = 0.88 in alc). Sol in alcohol, methanol, benzene, dil alkalies. Absorption spectra: Farmilo, Bull. Narc. 6, No. 3/4, 18 (1954).Derivative
Hydrobromide.C18H19NO3.HBr; mol wt 378.26.
C 57.15%, H 5.33%, N 3.70%, O 12.69%, Br 21.12%.
Properties
Crystals from water, mp 270-275°. Also an acid hydrobromide, (B.HBr)2.HBr.Derivative
Hydrochloride.C18H19NO3.HCl; mol wt 333.81.
C 64.77%, H 6.04%, N 4.20%, O 14.38%, Cl 10.62%.
Properties
Needles from water, mp 256-260°. Also an acid hydrochloride, (B.HCl)2.HCl. Absorption spectrum: Csokán, Z. Anal. Chem. 124, 344 (1942).Derivative
Hydriodide.C18H19NO3.HI; mol wt 425.26.
C 50.84%, H 4.74%, N 3.29%, O 11.29%, I 29.84%.
Properties
Clusters from dil alc, mp 243-244° (with preliminary softening at 237°).Derivative
Methiodide.C18H19NO3.CH3I; mol wt 439.29.
C 51.95%, H 5.05%, N 3.19%, O 10.93%, I 28.89%.
Properties
Crystals from acetic acid, mp 221-222°.Status
This monograph has been retired and is no longer subject to revision or update.