13-06955. Osajin [Archived]
Nomenclature
CAS number: 482-53-1
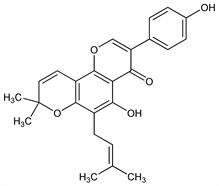
5-Hydroxy-3-(4-hydroxyphenyl)-8,8-dimethyl-6-(3-methyl-2-butenyl)-4H,8H-benzo[1,2-b:3,4-b′]dipyran-4-one. C25H24O5; mol wt 404.46.
C 74.24%, H 5.98%, O 19.78%.
Description and references
Isoln from osage oranges, Maclura pomifera (Raf.) Schneid. (M. aurantiaca Nutt.), Moraceae: Walter et al., J. Am. Chem. Soc. 60, 574 (1938); Wolfrom, Mahan, ibid. 64, 308 (1942). Structure: Wolfrom et al., ibid. 68, 406 (1946). Synthesis of osajin 4′-methyl ether: Jain et al., Tetrahedron 26, 1977 (1970). Synthesis of osajin: Jain, Sharma, J. Org. Chem. 39, 2215 (1974).
Properties
Light yellow crystals from xylene or 95% ethanol, mp 189° (uncorr), 193° (corr). Practically insol in water, petr ether, moderately sol in benzene, alcohol, warm carbon tetrachloride; very sol in chloroform, ether, acetone, pyridine. uv max (abs alc): 274 nm, Horowitz, Jurd, J. Org. Chem. 26, 2446 (1961).Status
This monograph has been retired and is no longer subject to revision or update.