13-09029. Sulfasomizole [Archived]
Nomenclature
CAS number: 632-00-8
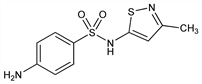
4-Amino-N-(3-methyl-5-isothiazolyl)benzenesulfonamide; 3-methyl-5-sulfanilamidoisothiazole; 5-p-aminobenzenesulfonamido-3-methylisothiazole; 5-sulfanilamido-3-methylisothiazole; N1-(3-methylisothiazol-5-yl)sulfanilamide; sulphasomizole; Bidizole; Amidozol. C10H11N3O2S2; mol wt 269.34.
C 44.59%, H 4.12%, N 15.60%, O 11.88%, S 23.81%.
Description and references
Preparation from N-acetylsulfanilyl chloride and 5-amino-3-methylisothiazole in pyridine followed by deacetylation in 2N NaOH: Adams, Slack, J. Chem. Soc. 1959, 3070; Adams et al., Nature 186, 221 (1960); GB 835753 (1960 to May & Baker).
Properties
Cream-colored needles from H2O, mp 192-192.5°. Soly in H2O at pH 6.0: 248 mg/100 ml; at pH 7.0: 2.26 g/100 ml.Derivative
Sodium salt monohydrate.C10H10N3NaO2S2.H2O; mol wt 309.34.
C 38.83%, H 3.91%, N 13.58%, Na 7.43%, O 15.52%, S 20.73%.
Properties
Crystals, when anhydr dec 345°. Freely sol in water. Aq solns of almost neutral pH contg more than 50% w/v of the sodium salt can be made.Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Synthetic); Sulfonamides
Status
This monograph has been retired and is no longer subject to revision or update.