13-09597. Tolonidine [Archived]
Nomenclature
Description and references
Orally active antihypertensive agent, related structurally to clonidine. Prepn: NL 6411516; US 3236857; US 6411516; US 3454701 (1965, 1966, 1969 all to Boehringer, Ing.). Synthesis and hypotensive activity: P. B. M. Timmermans et al., Rec. Trav. Chim. 97, 51 (1978). Pharmacological properties: C. Cosnier et al., Arzneim.-Forsch. 25, 1557, 1802, 1926 (1975). Structure-activity study: P. B. M. Timmermans, P.A. Van Zwieten, J. Med. Chem. 20, 1636 (1977). Chromatographic study: P. B. M. Timmermans et al., J. Chromatogr. 144, 215 (1977). Quantum chemical studies: eidem, Arzneim.-Forsch. 27, 2266 (1977). Radioimmunoassay disposition study: B. Jarrott, S. Spector, J. Pharmacol. Exp. Ther. 207, 195 (1978). Toxicity study: D. Cosnier et al., Arzneim.-Forsch. 25, 1926 (1975).
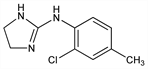
Properties
Crystals from benzene/petr ether, mp 148-150°. pKa 9.41.Derivative
Nitrate.Nomenclature
Properties
Crystals, mp 162-164°. LD50 in male mice, rats (mg/kg): 160, 420 orally; 21.25, 42 i.v. (Cosnier).Therapeutic Category
Antihypertensive.
Keywords
Antihypertensive; Imidazole Derivatives
Status
This monograph has been retired and is no longer subject to revision or update.