|
|
|
|
|
|
|
for choosing the right candidate drug with the ideal kinetic profile for the medical application to be treated prior to full development and for choosing the first and subsequent doses in man. Two methods are being investigated with increasing success using both in vitro and in vivo approaches. |
|
|
|
|
|
|
|
|
For almost 20 years, the possibility of predicting in vivo clearance from in vitro incubations has been discussed [40]. The more recent advances in in vitro metabolic methodology, together with the ability to identify and measure metabolites using HPLC linked with quadrapole MS-MS without the need to synthesize radioactive compounds, has dramatically improved the measurement of the rates of metabolism at an early stage in drug development. Recently, Houston [41] reviewed the available methods and, using published data, attempted to evaluate the accuracy of these predictions. There are 3 stages in the extrapolation from in vitro metabolism to whole body clearance (Figure 4). |
|
|
|
|
|
|
|
|
The first stage is to calculate the total in vitro metabolic rate, either by measurement of drug disappearancetaking into account other potential losses: volatilization, binding to apparatus, instability, etc.and/or by measurement of metabolic formation. This latter method, although preferable, is also more difficult since it must quantify all pathways. From rate curves, the maximum rate (Vm) and the Michaelis-Menten constant (Km) for drug-metabolizing enzyme binding are calculated according to Equation 1: |
|
|
|
|
|
|
|
|
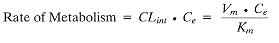 (1) (1) |
|
|
|
|
|
|
|
|
at low drug levels (<10% of Ce), and where CLint is the intrinsic clearance (a measure of the metabolizing capacity of the organ, independent of other factors such as absorption or protein binding), and Ce is the concentration of drug at the enzyme site. In practice, the intrinsic clearance is measured in terms of the total amount of enzyme protein present and this will depend on whether the incubations are using microsomes, hepatocytes, hepatic slices, etc. Thus the second stage is scaling up from these to the whole liver by using certain factors that are dependent on the initial protein. These values are subsequently incorporated in Equation 2, which relates intrinsic and hepatic clearance (CLh). |
|
|
|
|
|
|
|
|
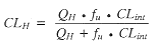 (2) (2) |
|
|
|
|
|
|
|
|
which uses the unbound free fraction in plasma (fu) and hepatic blood flow (QH) of the animal being investigated. Lastly, other routes of clearance, such as renal elimination, must be included wherever possible, although this may |
|
|
|
|
|