|
|
|
|
|
|
|
Unfortunately, not every novel research idea will translate into a commercially attractive product with a demonstrable therapeutic advantage. Innovation cannot be mandated, but it can be encouraged. A critical mass of scientific talent, coupled with a good communication network, relevant technologies, and an adequate infrastructure provides an environment that fosters innovative research. Challenging scientists to discover drugs of a unique chemical class, which modify biological processes in a novel way to improve or reverse a pathological condition in the patient, is one way to create a breakthrough product. So is encouraging innovative formulations and novel delivery systems. Genetic engineering techniques also promise to open up whole new dimensions of drug treatment by reconfiguring biological substances into therapeutic agents. |
|
|
|
|
|
|
|
|
A critical success factor for pharmaceutical companies in the future will be the ability to correctly identify these breakthrough products early in development. There is no proven method for predicting commercial success, but qualitative and quantitative analysis of the appropriate endpoints can be helpful. Quantitative analysis, such as determining the number of company patents that are cited as prior art by other patent applicants or clinical performance results, can be used to assess the value of the innovation [32]. Qualitative analysis, for example from an independent scientific advisory board who reviews reactions to papers presented at top scientific conferences or the value of publications, can be used to provide an unbiased assessment of the value of the company's research rationale and the technologies employed [33]. A plan for |
|
|
|
|
|
|
|
|
 |
|
|
|
|
|
|
|
|
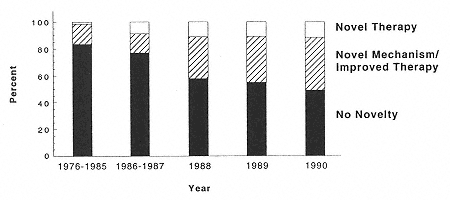
Fig. 4
Trend in innovative new project introductions. The graph shows a steady
increase in the fraction of new drug introductions that claim a novel mechanism
or therapeutic advance. (From Ref. [2].) |
|
|
|
|
|