| Entry |
| ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Name | Vigabatrin (JAN/USP/INN); Sabril (TN) | ||||||||||
| Product | SABRIL (Lundbeck Pharmaceuticals LLC), SABRIL (Lundbeck Pharmaceuticals LLC) | ||||||||||
| Generic | VIGABATRIN (Accord Healthcare), VIGABATRIN (Actavis Pharma), VIGABATRIN (Actavis Pharma), VIGABATRIN (Amneal Pharmaceuticals NY LLC), VIGABATRIN (Amneal Pharmaceuticals NY LLC), VIGABATRIN (Ascend Laboratories), VIGADRONE (Aucta Pharmaceuticals), VIGADRONE (Aucta Pharmaceuticals), VIGABATRIN (Aurobindo Pharma Limited), VIGABATRIN (Aurobindo Pharma Limited), VIGABATRIN (Biocon Pharma), VIGABATRIN (Cadila Healthcare Limited), VIGABATRIN FOR ORAL SOLUTION (Camber Pharmaceuticals), VIGABATRIN (Cipla USA), VIGABATRIN (Dr. Reddy's Laboratories Inc.,), VIGABATRIN (Dr. Reddy's Laboratories Inc.,), VIGABATRIN (Elite Laboratories), VIGABATRIN FOR ORAL SOLUTION USP, 500 MG (Granules Pharmaceuticals), VIGABATRIN FOR ORAL SOLUTION USP, 500 MG (Hikma Pharmaceuticals USA), VIGABATRIN (Lupin Pharmaceuticals), VIGABATRIN (Padagis US LLC), VIGABATRIN (Padagis US LLC), VIGABATRIN (Par Pharmaceutical), VIGADRONE (Upsher-Smith Laboratories), VIGABATRIN (Zydus Pharmaceuticals (USA)) | ||||||||||
| Formula | C6H11NO2 | ||||||||||
| Exact mass | 129.079 | ||||||||||
| Mol weight | 129.157 | ||||||||||
| Structure | 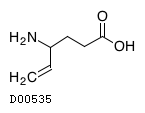 | ||||||||||
| Simcomp | |||||||||||
| Class | Neuropsychiatric agent DG03199 Antiepileptic agent DG02037 GABA mimetic antiepileptic | ||||||||||
| Remark |
| ||||||||||
| Efficacy | Anticonvulsant, Antiepileptic | ||||||||||
| Disease | Infantile spasms [DS:H01460] | ||||||||||
| Comment | Fatty acid derivative | ||||||||||
| Target | ABAT [HSA:18] [KO:K13524] | ||||||||||
| Pathway |
| ||||||||||
| Interaction | |||||||||||
| Structure map |
| ||||||||||
| Brite | Anatomical Therapeutic Chemical (ATC) classification [BR:br08303] N NERVOUS SYSTEM N03 ANTIEPILEPTICS N03A ANTIEPILEPTICS N03AG Fatty acid derivatives N03AG04 Vigabatrin D00535 Vigabatrin (JAN/USP/INN) <JP/US> USP drug classification [BR:br08302] Anticonvulsants Gamma-aminobutyric Acid (GABA) Augmenting Agents GABA Agents, Other Vigabatrin D00535 Vigabatrin (JAN/USP/INN) Therapeutic category of drugs in Japan [BR:br08301] 1 Agents affecting nervous system and sensory organs 11 Agents affecting central nervous system 113 Antiepileptics 1139 Others D00535 Vigabatrin (JAN/USP/INN) Drug groups [BR:br08330] Neuropsychiatric agent DG03199 Antiepileptic agent DG02037 GABA mimetic antiepileptic D00535 Vigabatrin Drug classes [BR:br08332] Neuropsychiatric agent DG03199 Antiepileptic agent D00535 Vigabatrin Target-based classification of drugs [BR:br08310] Enzymes Transferases (EC2) Aminotransferase ABAT D00535 Vigabatrin (JAN/USP/INN) <JP/US> New drug approvals in the USA [br08319.html] New molecular entities and new therapeutic biological products D00535 New drug approvals in Europe [br08329.html] European public assessment reports (EPAR) authorised medicine D00535 New drug approvals in Japan [br08318.html] Drugs with new active ingredients D00535 New drug approvals in the USA, Europe and Japan [br08328.html] Approval dates by FDA, EMA and PMDA D00535 | ||||||||||
| Other DBs |
| ||||||||||
| KCF data | ATOM 9 1 C1c C 17.4113 -16.5555 2 C1b C 18.6260 -15.8548 3 N1a N 16.1967 -15.8607 4 C2b C 17.4172 -17.9570 5 C1b C 19.8406 -16.5438 6 C6a C 21.0553 -15.8490 7 O6a O 22.2641 -16.5381 8 O6a O 21.0494 -14.4416 9 C2a C 16.2033 -18.6648 BOND 8 1 1 2 1 2 1 3 1 3 1 4 1 4 2 5 1 5 5 6 1 6 6 7 1 7 6 8 2 8 4 9 2 |