| Entry |
| ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Name | Dexamethasone sodium phosphate (JAN/USP); Dalalone (TN); Maxidex (TN) | ||||||||||
| Product | |||||||||||
| Generic | DEXAMETHASONE SODIUM PHOSPHATE (A-S Medication Solutions), DEXAMETHASONE SODIUM PHOSPHATE (A-S Medication Solutions), DEXAMETHASONE SODIUM PHOSPHATE (A-S Medication Solutions), DEXAMETHASONE SODIUM PHOSPHATE (A-S Medication Solutions), DEXAMETHASONE SODIUM PHOSPHATE (A-S Medication Solutions), DEXAMETHASONE SODIUM PHOSPHATE (Amneal Pharmaceuticals LLC), DEXAMETHASONE SODIUM PHOSPHATE (Amneal Pharmaceuticals LLC), DEXAMETHASONE SODIUM PHOSPHATE (Asclemed USA), DEXAMETHASONE SODIUM PHOSPHATE (Asclemed USA), DEXAMETHASONE SODIUM PHOSPHATE (AuroMedics Pharma LLC), DEXAMETHASONE SODIUM PHOSPHATE (AuroMedics Pharma LLC), DEXAMETHASONE SODIUM PHOSPHATE (AuroMedics Pharma LLC), DEXAMETHASONE SODIUM PHOSPHATE (Bausch & Lomb Incorporated), DEXAMETHASONE SODIUM PHOSPHATE (Cardinal Health), DEXAMETHASONE SODIUM PHOSPHATE (Cardinal Health), DEXAMETHASONE SODIUM PHOSPHATE (Cardinal Health), DEXAMETHASONE SODIUM PHOSPHATE (Civica), DEXAMETHASONE SODIUM PHOSPHATE (Fresenius Kabi USA), DEXAMETHASONE SODIUM PHOSPHATE (Fresenius Kabi USA), DEXAMETHASONE SODIUM PHOSPHATE (Fresenius Kabi USA), DEXAMETHASONE SODIUM PHOSPHATE (Fresenius Kabi USA), DEXAMETHASONE SODIUM PHOSPHATE (Fresenius Kabi USA), DEXAMETHASONE SODIUM PHOSPHATE (Fresenius Kabi USA), DEXAMETHASONE SODIUM PHOSPHATE (Fresenius Kabi USA), DEXAMETHASONE SODIUM PHOSPHATE (General Injectables & Vaccines), DEXAMETHASONE SODIUM PHOSPHATE (General Injectables and Vaccines), DEXAMETHASONE SODIUM PHOSPHATE (General Injectables and Vaccines), DEXAMETHASONE SODIUM PHOSPHATE (Gland Pharma Limited), DEXAMETHASONE SODIUM PHOSPHATE (HF Acquisition Co LLC), DEXAMETHASONE SODIUM PHOSPHATE (HF Acquisition Co LLC), DEXAMETHASONE SODIUM PHOSPHATE (HF Acquisition Co LLC), DEXAMETHASONE SODIUM PHOSPHATE (HF Acquisition Co LLC), DEXAMETHASONE SODIUM PHOSPHATE (HF Acquisition Co LLC), DEXAMETHASONE SODIUM PHOSPHATE (HF Acquisition Co LLC), DEXAMETHASONE SODIUM PHOSPHATE (Henry Schein), DEXAMETHASONE SODIUM PHOSPHATE (Henry Schein), DEXAMETHASONE SODIUM PHOSPHATE (Henry Schein), DEXAMETHASONE SODIUM PHOSPHATE (Hikma Pharmaceuticals USA), VIADOX (IT3 Medical LLC), CORTAREN CORTICOSTEROID/ANTI-INFLAMMATORY SYSTEM (IT3 Medical LLC), NEUROMAQUEL NEUROMA/ANTI-INFLAMMATORY SYSTEM (IT3 Medical LLC), DEXAMETHASONE SODIUM PHOSPHATE (Medical Purchasing Solutions), DEXAMETHASONE SODIUM PHOSPHATE (Medical Purchasing Solutions), DEXAMETHASONE SODIUM PHOSPHATE (Medical Purchasing Solutions), DEXAMETHASONE SODIUM PHOSPHATE (Medical Purchasing Solutions), DEXAMETHASONE SODIUM PHOSPHATE (Medical Purchasing Solutions), DEXAMETHASONE SODIUM PHOSPHATE (Medical Purchasing Solutions), DEXAMETHASONE SODIUM PHOSPHATE (Medical Purchasing Solutions), DEXAMETHASONE SODIUM PHOSPHATE (Medical Purchasing Solutions), DEXAMETHASONE SODIUM PHOSPHATE (Medical Purchasing Solutions), DEXAMETHASONE SODIUM PHOSPHATE (Medical Purchasing Solutions), DEXAMETHASONE SODIUM PHOSPHATE (Medical Purchasing Solutions), DEXAMETHASONE SODIUM PHOSPHATE (Mylan Institutional LLC), DEXAMETHASONE SODIUM PHOSPHATE (Mylan Institutional LLC), DEXAMETHASONE SODIUM PHOSPHATE (Phlow Corporation), DEXAMETHASONE SODIUM PHOSPHATE (REMEDYREPACK), DEXAMETHASONE SODIUM PHOSPHATE (REMEDYREPACK), DEXAMETHASONE SODIUM PHOSPHATE (REMEDYREPACK), DEXAMETHASONE SODIUM PHOSPHATE (REMEDYREPACK), DEXAMETHASONE SODIUM PHOSPHATE (Somerset Therapeutics), DEXAMETHASONE SODIUM PHOSPHATE (Somerset Therapeutics), DEXAMETHASONE SODIUM PHOSPHATE (Somerset Therapeutics) | ||||||||||
| Formula | C22H28FO8P. 2Na | ||||||||||
| Exact mass | 516.1301 | ||||||||||
| Mol weight | 516.4046 | ||||||||||
| Structure | 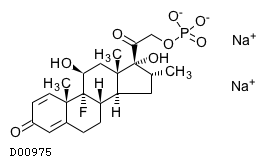 | ||||||||||
| Simcomp | |||||||||||
| Class | Anti-inflammatory DG01955 Corticosteroid DG02068 Glucocorticoid Metabolizing enzyme substrate DG01633 CYP3A/CYP3A4 substrate DG02913 CYP3A4 substrate Metabolizing enzyme inducer DG02853 CYP3A/CYP3A4 inducer DG01635 CYP3A4 inducer | ||||||||||
| Remark |
| ||||||||||
| Efficacy | Anti-inflammatory, Glucocorticoid receptor agonist | ||||||||||
| Disease | Primary or secondary adrenocortical insufficiency [DS:H01598] Congenital adrenal hyperplasia [DS:H00216] Rheumatoid arthritis [DS:H00630] Juvenile rheumatoid arthritis [DS:H01672] Acute gouty arthritis [DS:H01532] Psoriatic arthritis [DS:H01507] Ankylosing spondylitis [DS:H01674] Systemic lupus erythematosus [DS:H00080] Pemphigus [DS:H01648] Severe erythema multiforme [DS:H01695] Stevens-Johnson syndrome [DS:H01694] Seborrheic dermatitis [DS:H01652] Psoriasis [DS:H01656] Mycosis fungoides [DS:H01463] Bronchial asthma [DS:H00079] Contact dermatitis [DS:H01357] Atopic dermatitis [DS:H01358] Seasonal or perennial allergic rhinitis [DS:H01360] Herpes zoster ophthalmicus [DS:H00366] Optic neuritis [DS:H01717] Ulcerative colitis [DS:H01466] Pulmonary tuberculosis [DS:H00342] Acquired (autoimmune) hemolytic anemia [DS:H01585] Nephrotic syndrome [DS:H01657] | ||||||||||
| Target | NR3C1 (GR) [HSA:2908] [KO:K05771] | ||||||||||
| Pathway |
| ||||||||||
| Metabolism | Enzyme: CYP3A4 [HSA:1576] | ||||||||||
| Interaction | CYP induction: CYP3A4 [HSA:1576] | ||||||||||
| Structure map |
| ||||||||||
| Brite | Anatomical Therapeutic Chemical (ATC) classification [BR:br08303] A ALIMENTARY TRACT AND METABOLISM A01 STOMATOLOGICAL PREPARATIONS A01A STOMATOLOGICAL PREPARATIONS A01AC Corticosteroids for local oral treatment A01AC02 Dexamethasone D00975 Dexamethasone sodium phosphate (JAN/USP) <JP/US> C CARDIOVASCULAR SYSTEM C05 VASOPROTECTIVES C05A AGENTS FOR TREATMENT OF HEMORRHOIDS AND ANAL FISSURES FOR TOPICAL USE C05AA Corticosteroids C05AA09 Dexamethasone D00975 Dexamethasone sodium phosphate (JAN/USP) <JP/US> D DERMATOLOGICALS D07 CORTICOSTEROIDS, DERMATOLOGICAL PREPARATIONS D07A CORTICOSTEROIDS, PLAIN D07AB Corticosteroids, moderately potent (group II) D07AB19 Dexamethasone D00975 Dexamethasone sodium phosphate (JAN/USP) <JP/US> D07X CORTICOSTEROIDS, OTHER COMBINATIONS D07XB Corticosteroids, moderately potent, other combinations D07XB05 Dexamethasone D00975 Dexamethasone sodium phosphate (JAN/USP) <JP/US> D10 ANTI-ACNE PREPARATIONS D10A ANTI-ACNE PREPARATIONS FOR TOPICAL USE D10AA Corticosteroids, combinations for treatment of acne D10AA03 Dexamethasone D00975 Dexamethasone sodium phosphate (JAN/USP) <JP/US> H SYSTEMIC HORMONAL PREPARATIONS, EXCL. SEX HORMONES AND INSULINS H02 CORTICOSTEROIDS FOR SYSTEMIC USE H02A CORTICOSTEROIDS FOR SYSTEMIC USE, PLAIN H02AB Glucocorticoids H02AB02 Dexamethasone D00975 Dexamethasone sodium phosphate (JAN/USP) <JP/US> R RESPIRATORY SYSTEM R01 NASAL PREPARATIONS R01A DECONGESTANTS AND OTHER NASAL PREPARATIONS FOR TOPICAL USE R01AD Corticosteroids R01AD03 Dexamethasone D00975 Dexamethasone sodium phosphate (JAN/USP) <JP/US> S SENSORY ORGANS S01 OPHTHALMOLOGICALS S01B ANTIINFLAMMATORY AGENTS S01BA Corticosteroids, plain S01BA01 Dexamethasone D00975 Dexamethasone sodium phosphate (JAN/USP) <JP/US> S01C ANTIINFLAMMATORY AGENTS AND ANTIINFECTIVES IN COMBINATION S01CB Corticosteroids/antiinfectives/mydriatics in combination S01CB01 Dexamethasone D00975 Dexamethasone sodium phosphate (JAN/USP) <JP/US> S02 OTOLOGICALS S02B CORTICOSTEROIDS S02BA Corticosteroids S02BA06 Dexamethasone D00975 Dexamethasone sodium phosphate (JAN/USP) <JP/US> S03 OPHTHALMOLOGICAL AND OTOLOGICAL PREPARATIONS S03B CORTICOSTEROIDS S03BA Corticosteroids S03BA01 Dexamethasone D00975 Dexamethasone sodium phosphate (JAN/USP) <JP/US> USP drug classification [BR:br08302] Hormonal Agents, Stimulant/Replacement/Modifying (Adrenal) Dexamethasone D00975 Dexamethasone sodium phosphate (JAN/USP) Inflammatory Bowel Disease Agents Glucocorticoids Dexamethasone D00975 Dexamethasone sodium phosphate (JAN/USP) Ophthalmic Agents Ophthalmic Anti-inflammatories Glucocorticoids, Ophthalmic Dexamethasone D00975 Dexamethasone sodium phosphate (JAN/USP) Therapeutic category of drugs in Japan [BR:br08301] 1 Agents affecting nervous system and sensory organs 13 Agents affecting sensory organs 131 Ophthalmic agents 1315 Ophthalmic cortisones D00975 Dexamethasone sodium phosphate (JAN/USP) 132 Otic and nasal agents 1329 Others D00975 Dexamethasone sodium phosphate (JAN/USP) 2 Agents affecting individual organs 24 Hormones 245 Adrenal hormone preparations 2454 Fluorinated adrenocorticotropic hormones D00975 Dexamethasone sodium phosphate (JAN/USP) Drug groups [BR:br08330] Anti-inflammatory DG01955 Corticosteroid DG02068 Glucocorticoid DG00011 Dexamethasone D00975 Dexamethasone sodium phosphate Metabolizing enzyme substrate DG01633 CYP3A/CYP3A4 substrate DG02913 CYP3A4 substrate DG00011 Dexamethasone D00975 Dexamethasone sodium phosphate Metabolizing enzyme inducer DG02853 CYP3A/CYP3A4 inducer DG01635 CYP3A4 inducer DG00011 Dexamethasone D00975 Dexamethasone sodium phosphate Drug classes [BR:br08332] Anti-inflammatory DG01955 Corticosteroid D00975 Dexamethasone sodium phosphate Target-based classification of drugs [BR:br08310] Nuclear receptors Estrogen like receptors 3-Ketosteroid receptor NR3C1 (GR) D00975 Dexamethasone sodium phosphate (JAN/USP) <JP/US> Drug metabolizing enzymes and transporters [br08309.html] Drug metabolizing enzymes D00975 Drug groups [BR:br08330] Anti-inflammatory DG01955 Corticosteroid DG02068 Glucocorticoid DG00011 Dexamethasone Metabolizing enzyme substrate DG01633 CYP3A/CYP3A4 substrate DG02913 CYP3A4 substrate DG00011 Dexamethasone Metabolizing enzyme inducer DG02853 CYP3A/CYP3A4 inducer DG01635 CYP3A4 inducer DG00011 Dexamethasone | ||||||||||
| Other DBs |
| ||||||||||
| KCF data | ATOM 34 1 C1y C 20.5509 -18.6771 2 C1y C 19.3622 -19.3588 3 C1z C 20.5626 -17.2961 4 C1x C 22.9399 -18.7004 5 C1z C 18.1794 -18.6655 6 C1x C 19.3622 -20.7281 7 C1z C 21.7686 -16.6144 8 C1x C 19.3855 -16.6144 9 C1a C 20.5567 -15.9269 10 C1y C 22.9514 -17.3253 11 C1z C 16.9908 -19.3471 12 C1y C 18.1910 -17.2961 13 X F 18.1677 -20.0289 14 C1x C 18.1677 -21.4098 15 C5a C 21.7745 -15.2451 16 O1a O 23.0913 -16.2589 17 C1a C 24.2741 -17.1681 18 C2y C 16.9849 -20.7222 19 C2x C 15.8021 -18.6596 20 C1a C 16.9791 -17.9662 21 O1a O 17.0025 -16.6028 22 C1b C 22.9573 -14.5575 23 O5a O 20.5743 -14.5575 24 C2x C 15.8021 -21.4040 25 C2x C 14.6135 -19.3471 26 O2b O 24.1401 -15.2334 27 C5x C 14.6135 -20.7222 28 P1b P 25.2763 -14.4585 29 O5x O 13.4190 -21.3981 30 O1c O 26.4534 -15.1461 31 O1c O 26.4068 -13.6836 #- 32 O1c O 24.5014 -13.3223 #- 33 Z Na 28.9880 -14.9538 #+ 34 Z Na 28.9181 -18.1935 #+ BOND 35 1 1 2 1 2 1 3 1 3 1 4 1 4 2 5 1 5 2 6 1 6 3 7 1 7 3 8 1 8 3 9 1 #Up 9 4 10 1 10 5 11 1 11 5 12 1 12 5 13 1 #Down 13 6 14 1 14 7 15 1 #Up 15 7 16 1 #Down 16 10 17 1 #Down 17 11 18 1 18 11 19 1 19 11 20 1 #Up 20 12 21 1 #Up 21 15 22 1 22 15 23 2 23 18 24 2 24 19 25 2 25 22 26 1 26 24 27 1 27 26 28 1 28 27 29 2 29 28 30 2 30 28 31 1 31 28 32 1 32 7 10 1 33 8 12 1 34 14 18 1 35 25 27 1 |