13. Acacetin
Nomenclature
CAS number: 480-44-4
5,7-Dihydroxy-2-(4-methoxyphenyl)-4H-1-benzopyran-4-one; 5,7-dihydroxy-4′-methoxyflavone; apigenin-4′-methyl ether. C16H12O5; mol wt 284.26.
C 67.60%, H 4.26%, O 28.14%.
Description and references
The aglycon of linarin, q.v., and of acaciin. Isoln from linarin: Zemplén, Bognar, Ber. 74B, 1818 (1941). From acaciin: Hattori, Acta Phytochim. 2, 105 (1925). Isoln from Robinia pseudoacacia L., Leguminosae: Nakazawa, Matsuura, J. Pharm. Soc. Jpn. 73, 481 (1953). Structure: Baker et al., J. Chem. Soc. 1951, 691. Synthesis: Robinson, Venkataraman, ibid. 1926, 2348; Zemplén, Bognar, Ber. 76B, 452 (1943); Narasimhachari, Seshadri, Proc. Indian Acad. Sci. 30A, 151 (1949); Simpson, Sci. Proc. R. Dublin Soc. 27, 111 (1956), C.A. 51, 8082a (1957).
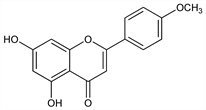
Properties
Yellow needles from 95% alcohol, mp 263°. Sol in hot alc, practically insol in ether. Sol in alkalies with yellow color.Derivative
7-Rhamnoglucoside.Nomenclature
Acaciin.C28H32O14; mol wt 592.55.
C 56.75%, H 5.44%, O 37.80%.
Description and references
From Robinia pseudacacia L., Leguminosae: Freudenberg, Hartmann, Ann. 587, 207 (1954). Structure: Zemplén, Mester, Magy. Kem. Foly. 56, 2 (1950), C.A. 45, 7977d (1951).