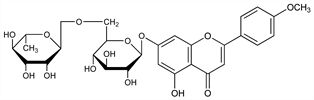
5498. Linarin
Nomenclature
CAS number: 480-36-4
7-[[6-O-(6-Deoxy-α-l-mannopyranosyl)-β-d-glucopyranosyl]oxy]-5-hydroxy-2-(4-methoxyphenyl)-4H-1-benzopyran-4-one; acacetin-β-rutinoside; linarigenin-glucoside; 5,7-dihydroxy-4′-methoxyflavone-d-glucosido-l-rhamnoside; buddleoflavonoloside. C28H32O14; mol wt 592.55.
C 56.75%, H 5.44%, O 37.80%.
Description and references
From the flowers of Linaria vulgaris Mill., Scrophulariaceae: Merz, Wu, Arch. Pharm. 274, 126 (1936); from Cirsium oleraceum Scop., Compositae: Wagner et al., ibid. 293, 1053 (1960). Structure: Baker et al., J. Chem. Soc. 1951, 691. Synthesis: Zemplén, Bognàr, Ber. 74, 1818 (1941).