172. Adrenochrome
Nomenclature
CAS number: 54-06-8
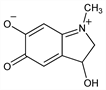
2,3-Dihydro-3-hydroxy-1-methyl-1H-indole-5,6-dione; 3-hydroxy-1-methyl-5,6-indolinedione. C9H9NO3; mol wt 179.17.
C 60.33%, H 5.06%, N 7.82%, O 26.79%.
Description and references
Pigment obtained by the oxidation of epinephrine. Prepn using Ag2O as oxidizing agent: MacCarthy, Chim. Ind. (Paris) 55, 435 (1946). Structure: D. E. Green, D. Richter, Biochem. J. 31, 596 (1937); J. Harley-Mason, Experientia 4, 307 (1948). Probably has the zwitterionic quinonimine structure shown here. Spectral data: A. Lund, Acta Pharmacol. Toxicol. 5, 1218 (1949). Review: R. A. Heacock, Chem. Rev. 59, 181-237 (1959). Review, as a psychotomimetic agent: R. A. Heacock, Chim. Ther. 6, 300 (1971).
Properties
Brilliant red crystals may exist as hemihydrate from methanol-formic acid, dec 115-120°. Absorption max (1 mg % aq sol): 220, 300, 485 nm (log ε 4.33, 4.01, 3.64). Well-formed and well-dried crystals can be kept in a vacuum desiccator for several weeks. Easily oxidized to melanin. Freely sol in water; fairly sol in alcohol. Almost insol in benzene and ether. Solns are unstable. Optimum pH of water soln 4.0.Derivative
Oxime sesquihydrate.Nomenclature
CAS number: 6055-73-8
C9H10N2O3.1H2O; mol wt 203.19.
C 53.20%, H 5.46%, N 13.79%, O 27.56%.
Properties
Orange needles from water, mp 278°. Much more stable than adrenochrome.Derivative
Monosemicarbazone.Nomenclature
CAS number: 69-81-8
Carbazochrome; Adrenoxyl (Sanofi Winthrop); Cromosil (Zambeletti). C10H12N4O3; mol wt 236.23.
C 50.84%, H 5.12%, N 23.72%, O 20.32%.
Description and references
Prepn: Dechamps et al., US 2506294 (1950 to Société Belge de l'Azote).
Properties
Orange-red crystals from dil alcohol, mp 203° (dec). See also Carbazochrome Salicylate.Derivative
Thiosemicarbazone.Nomenclature
CAS number: 113185-69-6
C10H12N4O2S; mol wt 252.29.
C 47.61%, H 4.79%, N 22.21%, O 12.68%, S 12.71%.
Description and references
Description: Fleischhacker, Barsel, US 2712024 (1955 to International Hormones).
Properties
mp 215-220°.Therapeutic Category
Monosemicarbazone as hemostatic.
Keywords
Hemostatic